External anions and cations distinguish between AMPA and kainate receptor gating mechanisms
- PMID: 11897844
- PMCID: PMC2290172
- DOI: 10.1113/jphysiol.2001.013407
External anions and cations distinguish between AMPA and kainate receptor gating mechanisms
Abstract
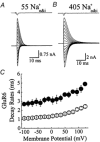
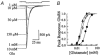
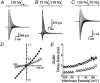
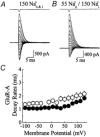
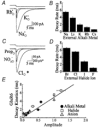
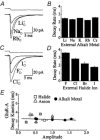
Experiments were designed to examine if ion-flow through alpha-amino-3-hydroxy-5-methyl-isoxazolepropionic acid (AMPA) or kainate receptors interferes with protein structures associated with the gating machinery. Gating was studied using ultra-fast drug perfusion of outside-out patches containing rat GluR-A or GluR6 subunits excised from transfected human embryonic kidney cells. Deactivation rates of GluR6 kainate receptors observed following brief L-glutamate (10 mM Glu, 1 ms) applications differed by two to threefold in high (405 mM symmetrical Na(+), tau(decay) = 2.7 ms at -100 mV) and low ionic strength (55 mM, tau(decay) = 1.1 ms) solutions. In comparison, GluR-A AMPA receptors were much less sensitive. Ion effects on GluR6 receptors did not reflect surface potential screening or ion-agonist competition at the agonist-binding site since deactivation rates were slower in high ionic strength solutions. Moreover, the apparent agonist affinity did not decrease with increasing ionic strength (e.g. 55 mM, EC(50) = 110 microM vs. 405 mM, EC(50) = 61 microM). GluR6 responses were strongly dependent on ions present on the external, but not the internal, side of the plasma membrane. Decay kinetics was regulated by the type of ion present suggesting that the chemical nature of the solution, not its ionic strength, governed channel behaviour. Both external anions and cations modulated the amplitude and decay kinetics of GluR6 responses in a concomitant manner. AMPA receptor responses recorded in identical ionic conditions did not exhibit this behaviour. These results identify a novel mechanism that distinguishes AMPA and kainate receptors. External ions regulate the gating machinery of kainate receptors through an allosteric mechanism that involves both anions and cations.
Figures
References
-
- Antonov SM, Gmiro VE, Johnson JW. Binding sites for permeant ions in the channel of NMDA receptors and their effects on channel block. Nature Neuroscience. 1998;1:451–456. - PubMed
-
- Arai A, Silberg J, Kessler M, Lynch G. Effect of thiocyanate on AMPA receptor mediated responses in excised patches and hippocampal slices. Neuroscience. 1995;66:815–827. - PubMed
-
- Armstrong N, Gouaux E. Mechanisms for activation and antagonism of an AMPA-sensitive glutamate receptor: crystal structures of the GluR2 ligand binding core. Neuron. 2000;28:165–181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

