Hydrogen peroxide increases depolarization-induced contraction of mechanically skinned slow twitch fibres from rat skeletal muscles
- PMID: 11897857
- PMCID: PMC2290173
- DOI: 10.1113/jphysiol.2001.013369
Hydrogen peroxide increases depolarization-induced contraction of mechanically skinned slow twitch fibres from rat skeletal muscles
Abstract
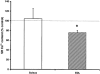
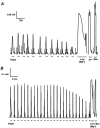
The effect of exogenous hydrogen peroxide (H(2)O(2)) on excitation-contraction (E-C) coupling and sarcoplasmic reticulum (SR) function was compared in mechanically skinned slow twitch fibres (prepared from the soleus muscles) and fast twitch fibres (prepared from the extensor digitorum longus; EDL muscles) of adult rats. Equilibration (5 min) with 1 mM H(2)O(2) diminished the ability of the Ca(2+)-depleted SR to reload Ca(2+) in both slow (P < 0.01) and fast twitch fibres (P < 0.05) compared to control. Under conditions when all Ca(2+) uptake was prevented, 1 mM H(2)O(2) increased SR Ca(2+) "leak" in fast twitch fibres by 24 +/- 5 % (P < 0.05), but leak was not altered in slow twitch fibres. Treatment with 1 mM H(2)O(2) also increased the peak force of low [caffeine] contracture by approximately 45% in both fibre types compared to control (P < 0.01), which could be partly reversed following treatment with 10 mM dithiothreitol (DTT). The changes in SR function caused by 1 mM H(2)O(2) were associated with an approximately 65% increase in the peak height of depolarization-induced contractile response (DICR) in slow twitch fibres, compared to control (no H(2)O(2); P < 0.05). In contrast, peak contractile force of fast twitch fibres was not altered by 1 mM H(2)O(2) treatment. Equilibration with 5 mM H(2)O(2) induced a spontaneous force response in both slow and fast twitch fibres, which could be partly reversed by 2 min treatment with 10 mM DTT. Peak DICR was also increased approximately 40% by 5 mM H(2)O(2) in slow twitch fibres compared to control (no H(2)O(2); P < 0.05). Our results indicate that exogenous H(2)O(2) increases depolarization-induced contraction of mechanically skinned slow but not fast twitch fibres. The increase in depolarization-induced contraction in slow twitch fibres might be mediated by an increased SR Ca(2+) release during contraction and/or an increase in Ca(2+) sensitivity.
Figures




Similar articles
-
Excitation-contraction coupling and sarcoplasmic reticulum function in mechanically skinned fibres from fast skeletal muscles of aged mice.J Physiol. 2002 Aug 15;543(Pt 1):169-76. doi: 10.1113/jphysiol.2002.022418. J Physiol. 2002. PMID: 12181289 Free PMC article.
-
Redox modulation of maximum force production of fast-and slow-twitch skeletal muscles of rats and mice.J Appl Physiol (1985). 2001 Mar;90(3):832-8. doi: 10.1152/jappl.2001.90.3.832. J Appl Physiol (1985). 2001. PMID: 11181590
-
Hydrogen peroxide modulates Ca2+-activation of single permeabilized fibres from fast- and slow-twitch skeletal muscles of rats.J Muscle Res Cell Motil. 2000;21(8):747-52. doi: 10.1023/a:1010344008224. J Muscle Res Cell Motil. 2000. PMID: 11392556
-
Effects of age on enzyme-histochemical fibre spectra and contractile properties of fast- and slow-twitch skeletal muscles in the rat.J Neurol Sci. 1986 Nov;76(1):69-89. doi: 10.1016/0022-510x(86)90143-7. J Neurol Sci. 1986. PMID: 2946814 Review.
-
Intracellular calcium movements during excitation-contraction coupling in mammalian slow-twitch and fast-twitch muscle fibers.J Gen Physiol. 2012 Apr;139(4):261-72. doi: 10.1085/jgp.201210773. J Gen Physiol. 2012. PMID: 22450485 Free PMC article. Review.
Cited by
-
Mitochondria-derived ROS bursts disturb Ca²⁺ cycling and induce abnormal automaticity in guinea pig cardiomyocytes: a theoretical study.Am J Physiol Heart Circ Physiol. 2015 Mar 15;308(6):H623-36. doi: 10.1152/ajpheart.00493.2014. Epub 2014 Dec 24. Am J Physiol Heart Circ Physiol. 2015. PMID: 25539710 Free PMC article.
-
Redox modulation of diaphragm contractility: Interaction between DHPR and RyR channels.Free Radic Biol Med. 2010 Dec 15;49(12):1969-77. doi: 10.1016/j.freeradbiomed.2010.09.025. Epub 2010 Oct 19. Free Radic Biol Med. 2010. PMID: 20920578 Free PMC article.
-
Excitation-contraction coupling and fatigue mechanisms in skeletal muscle: studies with mechanically skinned fibres.J Muscle Res Cell Motil. 2002;23(1):81-91. doi: 10.1023/a:1019932730457. J Muscle Res Cell Motil. 2002. PMID: 12363289 Review.
-
Calcium and Reactive Oxygen Species Signaling Interplays in Cardiac Physiology and Pathologies.Antioxidants (Basel). 2023 Feb 2;12(2):353. doi: 10.3390/antiox12020353. Antioxidants (Basel). 2023. PMID: 36829912 Free PMC article. Review.
-
Dual action of hydrogen peroxide on synaptic transmission at the frog neuromuscular junction.J Physiol. 2003 Oct 1;552(Pt 1):283-93. doi: 10.1113/jphysiol.2003.050690. Epub 2003 Aug 1. J Physiol. 2003. PMID: 12897166 Free PMC article.
References
-
- Abramson JJ, Salama G. Critical sulfhydryls regulate calcium release from sarcoplasmic reticulum. Journal of Bioenergetics and Biomembranes. 1989;21:283–294. - PubMed
-
- Armstrong RB, Phelps RO. Muscle fibre type composition of the rat hindlimb. American Journal of Anatomy. 1984;171:259–272. - PubMed
-
- Bakker AJ, Head SI, Wareham AC, Stephenson DG. Effect of clenbuterol on sarcoplasmic reticulum function in single skinned mammalian skeletal muscle fibres. American Journal of Physiology. 1998;274:C1718–1726. - PubMed
-
- Boraso A, Williams AJ. Modification of the gating of the cardiac sarcoplasmic reticulum Ca2+-release channel by H2O2 and dithiothreitol. American Journal of Physiology. 1994;267:H1010–1016. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

