The role of cytochrome c in caspase activation in Drosophila melanogaster cells
- PMID: 11901173
- PMCID: PMC2173478
- DOI: 10.1083/jcb.200111107
The role of cytochrome c in caspase activation in Drosophila melanogaster cells
Abstract
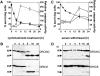
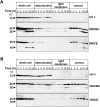
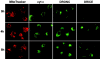
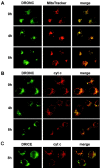
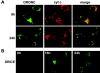
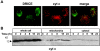
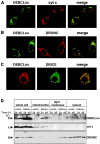
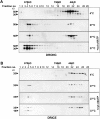
The release of cytochrome c from mitochondria is necessary for the formation of the Apaf-1 apoptosome and subsequent activation of caspase-9 in mammalian cells. However, the role of cytochrome c in caspase activation in Drosophila cells is not well understood. We demonstrate here that cytochrome c remains associated with mitochondria during apoptosis of Drosophila cells and that the initiator caspase DRONC and effector caspase DRICE are activated after various death stimuli without any significant release of cytochrome c in the cytosol. Ectopic expression of the proapoptotic Bcl-2 protein, DEBCL, also fails to show any cytochrome c release from mitochondria. A significant proportion of cellular DRONC and DRICE appears to localize near mitochondria, suggesting that an apoptosome may form in the vicinity of mitochondria in the absence of cytochrome c release. In vitro, DRONC was recruited to a >700-kD complex, similar to the mammalian apoptosome in cell extracts supplemented with cytochrome c and dATP. These results suggest that caspase activation in insects follows a more primitive mechanism that may be the precursor to the caspase activation pathways in mammals.
Figures

References
-
- Baehrecke, E.H. 2000. Steroid regulation of programmed cell death during Drosophila development. Cell Death Differ. 7:1057–1062. - PubMed
-
- Brachmann, C.B., O.W. Jassim, B.D. Wachsmuth, and R.L. Cagan. 2000. The Drosophila bcl-2 family member dBorg-1 functions in the apoptotic response to UV-irradiation. Curr. Biol. 10:547–550. - PubMed
-
- Cain, K., D.G. Brown, C. Langlais, and G.M. Cohen. 1999. Caspase activation involves the formation of the aposome, a large (∼700kDa) caspase-activating complex. J. Biol. Chem. 274:22686–22692. - PubMed
-
- Cain, K., S.B. Bratton, C. Langlais, G. Walker, D.G. Brown, X.-M. Sun, and G.M. Cohen. 2000. Apaf-1 oligomerises into biologically active ∼700-kDa and inactive ∼1.4MDa apoptosome complexes. J. Biol. Chem. 275:6067–6070. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

