Critical regions of secM that control its translation and secretion and promote secretion-specific secA regulation
- PMID: 11948148
- PMCID: PMC134986
- DOI: 10.1128/JB.184.9.2360-2369.2002
Critical regions of secM that control its translation and secretion and promote secretion-specific secA regulation
Abstract
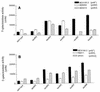
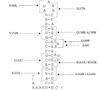
SecA is an essential ATP-driven motor protein that binds to presecretory or membrane proteins and the translocon and promotes the translocation or membrane integration of these proteins. secA is subject to a protein secretion-specific form of regulation, whereby its translation is elevated during secretion-limiting conditions. A novel mechanism that promotes this regulation involves translational pausing within the gene upstream of secA, secM. The secM translational pause prevents formation of an RNA helix that normally blocks secA translational initiation. The duration of this pause is controlled by the rate of secretion of nascent SecM, which in turn depends on its signal peptide and a functional translocon. We characterized the atypical secM signal peptide and found that mutations within the amino-terminal region specifically affect the secM translational pause and secA regulation, while mutations in the hydrophobic core region affect SecM secretion as well as translational pausing and secA regulation. In addition, mutational analysis of the 3' end of secM allowed us to identify a conserved region that is required to promote the translational pause that appears to be operative at the peptide level. Together, our results provide direct support for the secM translational pause model of secA regulation, and they pinpoint key sequences within secM that promote this important regulatory system.
Figures



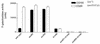
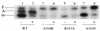

Similar articles
-
Translocon "pulling" of nascent SecM controls the duration of its translational pause and secretion-responsive secA regulation.J Bacteriol. 2003 Nov;185(22):6719-22. doi: 10.1128/JB.185.22.6719-6722.2003. J Bacteriol. 2003. PMID: 14594848 Free PMC article.
-
Revised translation start site for secM defines an atypical signal peptide that regulates Escherichia coli secA expression.J Bacteriol. 2000 Oct;182(19):5592-5. doi: 10.1128/JB.182.19.5592-5595.2000. J Bacteriol. 2000. PMID: 10986266 Free PMC article.
-
Translation arrest of SecM is essential for the basal and regulated expression of SecA.Proc Natl Acad Sci U S A. 2004 Aug 17;101(33):12330-5. doi: 10.1073/pnas.0404907101. Epub 2004 Aug 9. Proc Natl Acad Sci U S A. 2004. PMID: 15302932 Free PMC article.
-
Control of SecA and SecM translation by protein secretion.Curr Opin Microbiol. 2004 Apr;7(2):145-50. doi: 10.1016/j.mib.2004.01.001. Curr Opin Microbiol. 2004. PMID: 15063851 Review.
-
SecA protein: autoregulated ATPase catalysing preprotein insertion and translocation across the Escherichia coli inner membrane.Mol Microbiol. 1993 Jan;7(2):159-65. doi: 10.1111/j.1365-2958.1993.tb01107.x. Mol Microbiol. 1993. PMID: 8446024 Review.
Cited by
-
Translocon "pulling" of nascent SecM controls the duration of its translational pause and secretion-responsive secA regulation.J Bacteriol. 2003 Nov;185(22):6719-22. doi: 10.1128/JB.185.22.6719-6722.2003. J Bacteriol. 2003. PMID: 14594848 Free PMC article.
-
The Sec System: Protein Export in Escherichia coli.EcoSal Plus. 2017 Nov;7(2):10.1128/ecosalplus.ESP-0002-2017. doi: 10.1128/ecosalplus.ESP-0002-2017. EcoSal Plus. 2017. PMID: 29165233 Free PMC article. Review.
-
A nascent polypeptide domain that can regulate translation elongation.Proc Natl Acad Sci U S A. 2004 Mar 23;101(12):4059-64. doi: 10.1073/pnas.0400554101. Epub 2004 Mar 12. Proc Natl Acad Sci U S A. 2004. PMID: 15020769 Free PMC article.
-
Mechanisms of ribosome stalling by SecM at multiple elongation steps.Elife. 2015 Dec 14;4:e09684. doi: 10.7554/eLife.09684. Elife. 2015. PMID: 26670735 Free PMC article.
-
SecM facilitates translocase function of SecA by localizing its biosynthesis.Genes Dev. 2005 Feb 15;19(4):436-44. doi: 10.1101/gad.1259505. Genes Dev. 2005. PMID: 15713839 Free PMC article.
References
-
- Akita, M., S. Sasaki, S. Matsuyama, and S. Mizushima. 1990. SecA interacts with secretory proteins by recognizing the positive charge at the amino terminus of the signal peptide in Escherichia coli. J. Biol. Chem. 265:8164-8169. - PubMed
-
- Bankatis, V., B. Rasmussen, and P. J. Bassford. 1984. Intragenic suppressor mutations that restore export of maltose binding protein with a truncated signal peptide. Cell 37:243-252. - PubMed
-
- Bost, S., and D. Belin. 1997. prl mutations in the Escherichia coli secG gene. J. Biol. Chem. 272:4087-4093. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

