Endotoxin of Neisseria meningitidis composed only of intact lipid A: inactivation of the meningococcal 3-deoxy-D-manno-octulosonic acid transferase
- PMID: 11948150
- PMCID: PMC134985
- DOI: 10.1128/JB.184.9.2379-2388.2002
Endotoxin of Neisseria meningitidis composed only of intact lipid A: inactivation of the meningococcal 3-deoxy-D-manno-octulosonic acid transferase
Abstract
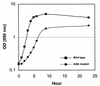
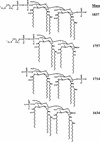
Lipopolysaccharide, lipooligosaccharide (LOS), or endotoxin is important in bacterial survival and the pathogenesis of gram-negative bacteria. A necessary step in endotoxin biosynthesis is 3-deoxy-D-manno-octulosonic acid (Kdo) glycosylation of lipid A, catalyzed by the Kdo transferase KdtA (WaaA). In enteric gram-negative bacteria, this step is essential for survival. A nonpolar kdtA::aphA-3 mutation was created in Neisseria meningitidis via allelic exchange, and the mutant was viable. Detailed structural analysis demonstrated that the endotoxin of the kdtA::aphA-3 mutant was composed of fully acylated lipid A with variable phosphorylation but without Kdo glycosylation. In contrast to what happens in other gram-negative bacteria, tetra-acylated lipid IV(A) did not accumulate. The LOS structure of the kdtA::aphA-3 mutant was restored to the wild-type structure by complementation with kdtA from N. meningitidis or Escherichia coli. The expression of a fully acylated, unglycosylated lipid A indicates that lipid A biosynthesis in N. meningitidis can proceed without the addition of Kdo and that KdtA is not essential for survival of the meningococcus.
Figures





Similar articles
-
KpsF is the arabinose-5-phosphate isomerase required for 3-deoxy-D-manno-octulosonic acid biosynthesis and for both lipooligosaccharide assembly and capsular polysaccharide expression in Neisseria meningitidis.J Biol Chem. 2002 Jul 5;277(27):24103-13. doi: 10.1074/jbc.M200931200. Epub 2002 Apr 15. J Biol Chem. 2002. PMID: 11956197
-
Inhibition of lipopolysaccharide biosynthesis and cell growth following inactivation of the kdtA gene in Escherichia coli.J Biol Chem. 1995 Nov 17;270(46):27646-52. doi: 10.1074/jbc.270.46.27646. J Biol Chem. 1995. PMID: 7499229
-
A novel 3-deoxy-D-manno-octulosonic acid (Kdo) hydrolase that removes the outer Kdo sugar of Helicobacter pylori lipopolysaccharide.J Bacteriol. 2005 May;187(10):3374-83. doi: 10.1128/JB.187.10.3374-3383.2005. J Bacteriol. 2005. PMID: 15866922 Free PMC article.
-
Genetic basis for biosynthesis, structure, and function of meningococcal lipooligosaccharide (endotoxin).Crit Rev Microbiol. 1998;24(4):281-334. doi: 10.1080/10408419891294216. Crit Rev Microbiol. 1998. PMID: 9887366 Review.
-
The atypical lipopolysaccharide of Francisella.Carbohydr Res. 2013 Aug 30;378:79-83. doi: 10.1016/j.carres.2013.06.015. Epub 2013 Jul 2. Carbohydr Res. 2013. PMID: 23916469 Free PMC article. Review.
Cited by
-
Meningococcal core and accessory phasomes vary by clonal complex.Microb Genom. 2020 May;6(5):e000367. doi: 10.1099/mgen.0.000367. Epub 2020 Apr 29. Microb Genom. 2020. PMID: 32375989 Free PMC article.
-
Toad's survivability and soil microbiome alterations impacted via individual abundance.Biol Futur. 2025 Sep;76(3):399-411. doi: 10.1007/s42977-025-00261-7. Epub 2025 Jun 6. Biol Futur. 2025. PMID: 40478395
-
Identification of a 3-deoxy-D-manno-octulosonic acid biosynthetic operon in Moraxella catarrhalis and analysis of a KdsA-deficient isogenic mutant.Infect Immun. 2003 Nov;71(11):6426-34. doi: 10.1128/IAI.71.11.6426-6434.2003. Infect Immun. 2003. PMID: 14573664 Free PMC article.
-
Adaptive genetic variation at three loci in South African vervet monkeys (Chlorocebus pygerythrus) and the role of selection within primates.PeerJ. 2018 Jun 4;6:e4953. doi: 10.7717/peerj.4953. eCollection 2018. PeerJ. 2018. PMID: 29888138 Free PMC article.
-
Identification of two late acyltransferase genes responsible for lipid A biosynthesis in Moraxella catarrhalis.FEBS J. 2008 Oct;275(20):5201-14. doi: 10.1111/j.1742-4658.2008.06651.x. Epub 2008 Sep 13. FEBS J. 2008. PMID: 18795947 Free PMC article.
References
-
- Belunis, C. J., T. Clementz, S. M. Carty, and C. R. Raetz. 1995. Inhibition of lipopolysaccharide biosynthesis and cell growth following inactivation of the kdtA gene in Escherichia coli. J. Biol. Chem. 270:27646-27652. - PubMed
-
- Belunis, C. J., K. E. Mdluli, C. R. Raetz, and F. E. Nano. 1992. A novel 3-deoxy-d-manno-octulosonic acid transferase from Chlamydia trachomatis required for expression of the genus-specific epitope. J. Biol. Chem. 267:18702-18707. - PubMed
-
- Belunis, C. J., and C. R. Raetz. 1992. Biosynthesis of endotoxins. Purification and catalytic properties of 3-deoxy-d-manno-octulosonic acid transferase from Escherichia coli. J. Biol. Chem. 267:9988-9997. - PubMed
-
- Bhat, U. R., L. S. Forsberg, and R. W. Carlson. 1994. Structure of lipid A component of Rhizobium leguminosarum bv. phaseoli lipopolysaccharide. Unique nonphosphorylated lipid A containing 2-amino-2-deoxygluconate, galacturonate, and glucosamine. J. Biol. Chem. 269:14402-14410. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

