Comprehensive human genome amplification using multiple displacement amplification
- PMID: 11959976
- PMCID: PMC122757
- DOI: 10.1073/pnas.082089499
Comprehensive human genome amplification using multiple displacement amplification
Abstract
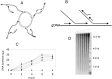
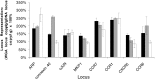
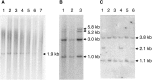
Fundamental to most genetic analysis is availability of genomic DNA of adequate quality and quantity. Because DNA yield from human samples is frequently limiting, much effort has been invested in developing methods for whole genome amplification (WGA) by random or degenerate oligonucleotide-primed PCR. However, existing WGA methods like degenerate oligonucleotide-primed PCR suffer from incomplete coverage and inadequate average DNA size. We describe a method, termed multiple displacement amplification (MDA), which provides a highly uniform representation across the genome. Amplification bias among eight chromosomal loci was less than 3-fold in contrast to 4-6 orders of magnitude for PCR-based WGA methods. Average product length was >10 kb. MDA is an isothermal, strand-displacing amplification yielding about 20-30 microg product from as few as 1-10 copies of human genomic DNA. Amplification can be carried out directly from biological samples including crude whole blood and tissue culture cells. MDA-amplified human DNA is useful for several common methods of genetic analysis, including genotyping of single nucleotide polymorphisms, chromosome painting, Southern blotting and restriction fragment length polymorphism analysis, subcloning, and DNA sequencing. MDA-based WGA is a simple and reliable method that could have significant implications for genetic studies, forensics, diagnostics, and long-term sample storage.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

