Comprehensive screening for human immunodeficiency virus type 1 subtype-specific CD8 cytotoxic T lymphocytes and definition of degenerate epitopes restricted by HLA-A0207 and -C(W)0304 alleles
- PMID: 11967314
- PMCID: PMC136178
- DOI: 10.1128/jvi.76.10.4971-4986.2002
Comprehensive screening for human immunodeficiency virus type 1 subtype-specific CD8 cytotoxic T lymphocytes and definition of degenerate epitopes restricted by HLA-A0207 and -C(W)0304 alleles
Abstract
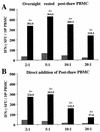
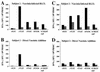
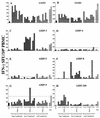
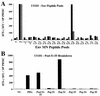
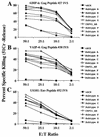
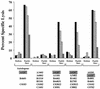
For this report, the rapid identification and characterization of human immunodeficiency virus type 1 (HIV-1)-derived broadly cross-subtype-reactive CD8 cytotoxic T lymphocyte (CTL) epitopes were performed. Using a gamma interferon (IFN-gamma) Elispot assay-based approach and a panel of recombinant vaccinia viruses expressing gag, env, pol, and nef genes representing the seven most predominant subtypes and one circulating recombinant form of HIV-1, the subtype specificity and cross-subtype reactivity of a CD8 response were directly measured from circulating peripheral blood mononuclear cells (PBMC). Enhanced sensitivity of detection of CD8 responses from cryopreserved PBMC was achieved using autologous vaccinia virus-infected B-lymphoblastoid cell lines as supplemental antigen-presenting cells. Of eleven subjects studied, six exhibited broadly cross-subtype-reactive CD8-mediated IFN-gamma production (at least seven of eight subtypes recognized) to at least one major gene product from HIV-1. Screening of subjects showing broadly cross-subtype-specific responses in the vaccinia virus-based enzyme-linked immunospot (Elispot) assay using a panel of overlapping peptides resulted in the identification of cross-subtype responses down to the 20-mer peptide level in less than 3 days. Three subjects showed broad cross-subtype reactivity in both the IFN-gamma Elispot assay and the standard chromium release cytotoxicity assay. Fine mapping and HLA restriction analysis of the response from three subjects demonstrated that this technique can be used to define epitopes restricted by HLA-A, -B, and -C alleles. In addition, the ability of all three epitopes to be processed from multiple subtypes of their parent proteins and presented in the context of HLA class I molecules following de novo synthesis is shown. While all three minimal epitopes mapped here had previously been defined as HIV-1 epitopes, two are shown to have novel HLA restriction alleles and therefore exhibit degenerate HLA binding capacity. These findings provide biological validation of HLA supertypes in HIV-1 CTL recognition and support earlier studies of cross-subtype CTL responses during HIV-1 infection.
Figures





Similar articles
-
CTL epitope distribution patterns in the Gag and Nef proteins of HIV-1 from subtype A infected subjects in Kenya: use of multiple peptide sets increases the detectable breadth of the CTL response.BMC Immunol. 2006 Apr 18;7:8. doi: 10.1186/1471-2172-7-8. BMC Immunol. 2006. PMID: 16620386 Free PMC article.
-
Cross-clade envelope glycoprotein 160-specific CD8+ cytotoxic T lymphocyte responses in early HIV type 1 clade B infection.AIDS Res Hum Retroviruses. 1998 Jul 20;14(11):925-37. doi: 10.1089/aid.1998.14.925. AIDS Res Hum Retroviruses. 1998. PMID: 9686639
-
An HLA-directed molecular and bioinformatics approach identifies new HLA-A11 HIV-1 subtype E cytotoxic T lymphocyte epitopes in HIV-1-infected Thais.AIDS Res Hum Retroviruses. 2001 May 20;17(8):703-17. doi: 10.1089/088922201750236988. AIDS Res Hum Retroviruses. 2001. PMID: 11429111
-
Identification of highly conserved and broadly cross-reactive HIV type 1 cytotoxic T lymphocyte epitopes as candidate immunogens for inclusion in Mycobacterium bovis BCG-vectored HIV vaccines.AIDS Res Hum Retroviruses. 2000 Sep 20;16(14):1433-43. doi: 10.1089/08892220050140982. AIDS Res Hum Retroviruses. 2000. PMID: 11018863 Review.
-
A systematic review of T cell epitopes defined from the proteome of human immunodeficiency virus.Virus Res. 2025 Aug;358:199602. doi: 10.1016/j.virusres.2025.199602. Epub 2025 Jun 23. Virus Res. 2025. PMID: 40562176 Free PMC article. Review.
Cited by
-
Hepatitis B virus (HBV)-specific T-cell responses to recombinant HBV core protein in patients with normal liver function and co-infected with chronic HBV and human immunodeficiency virus 1 (HIV-1).Virol J. 2013 Jul 12;10:232. doi: 10.1186/1743-422X-10-232. Virol J. 2013. PMID: 23849342 Free PMC article.
-
Limited breadth of a T-helper cell response to a human immunodeficiency virus envelope protein.J Virol. 2003 Apr;77(7):4231-6. doi: 10.1128/jvi.77.7.4231-4236.2003. J Virol. 2003. PMID: 12634380 Free PMC article.
-
The immunodominant influenza A virus M158-66 cytotoxic T lymphocyte epitope exhibits degenerate class I major histocompatibility complex restriction in humans.J Virol. 2014 Sep;88(18):10613-23. doi: 10.1128/JVI.00855-14. Epub 2014 Jul 2. J Virol. 2014. PMID: 24990997 Free PMC article.
-
CTL epitope distribution patterns in the Gag and Nef proteins of HIV-1 from subtype A infected subjects in Kenya: use of multiple peptide sets increases the detectable breadth of the CTL response.BMC Immunol. 2006 Apr 18;7:8. doi: 10.1186/1471-2172-7-8. BMC Immunol. 2006. PMID: 16620386 Free PMC article.
-
A double-blind randomized phase I clinical trial targeting ALVAC-HIV vaccine to human dendritic cells.PLoS One. 2011;6(9):e24254. doi: 10.1371/journal.pone.0024254. Epub 2011 Sep 16. PLoS One. 2011. PMID: 21949699 Free PMC article. Clinical Trial.
References
-
- Allen, T. M., D. H. O'Connor, P. Jing, J. L. Dzuris, B. R. Mothe, T. U. Vogel, E. Dunphy, M. E. Liebl, C. Emerson, N. Wilson, K. J. Kunstman, X. Wang, D. B. Allison, A. L. Hughes, R. C. Desrosiers, J. D. Altman, S. M. Wolinsky, A. Sette, and D. I. Watkins. 2000. Tat-specific cytotoxic T lymphocytes select for SIV escape variants during resolution of primary viraemia. Nature 407:386-390. - PubMed
-
- Altfeld, M. A., B. Livingston, N. Reshamwala, P. T. Nguyen, M. M. Addo, A. Shea, M. Newman, J. Fikes, J. Sidney, P. Wentworth, R. Chesnut, R. L. Eldridge, E. S. Rosenberg, G. K. Robbins, C. Brander, P. E. Sax, S. Boswell, T. Flynn, S. Buchbinder, P. J. Goulder, B. D. Walker, A. Sette, and S. A. Kalams. 2001. Identification of novel HLA-A2-restricted human immunodeficiency virus type 1-specific cytotoxic T-lymphocyte epitopes predicted by the HLA-A2 supertype peptide-binding motif. J. Virol. 75:1301-1311. - PMC - PubMed
-
- Artenstein, A. W., T. C. VanCott, J. R. Mascola, J. K. Carr, P. A. Hegerich, J. Gaywee, E. Sanders-Buell, M. L. Robb, D. E. Dayhoff, S. Thitivichianlert, S. Nitayaphan, J. G. McNeil, D. L. Birx, R. A. Michael, D. S. Burke, and F. E. McCutchan. 1995. Dual infection with human immunodeficiency virus type 1 of distinct envelope subtypes in humans. J. Infect. Dis. 171:805-810. - PubMed
-
- Bergmann, C. C., Q. Yao, C. K. Ho, and S. L. Buckwold. 1996. Flanking residues alter antigenicity and immunogenicity of multi-unit CTL epitopes. J. Immunol. 157:3242-3249. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

