Defining a T-cell epitope within HSP 65 in recurrent aphthous stomatitis
- PMID: 11985522
- PMCID: PMC1906391
- DOI: 10.1046/j.1365-2249.2002.01757.x
Defining a T-cell epitope within HSP 65 in recurrent aphthous stomatitis
Abstract
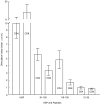
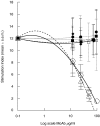
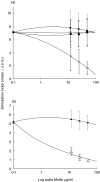
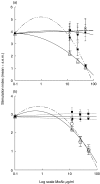
The 65 kD heat shock protein (HSP) has been implicated in the aetiology of recurrent aphthous stomatitis (RAS). We have previously demonstrated that peptide 91-105 derived from the sequence of mycobacterial 65 kD HSP stimulates specifically lymphocytes from patients with RAS. In this investigation, we show that both CD4+ and CD8+ T cells were significantly stimulated with mycobacterial peptide 91-105. In contrast, the human homologous peptide 116-130 stimulated only CD4+ T cells. Inhibition studies showed that CD4+ T cells were class II restricted, whereas CD8+ T cells were class I restricted. We then used truncated or substituted peptides, and demonstrated that residues 95-105 appear to be important, and residue 104(Arg) critical, in stimulating the T cells. Thus, peptide 95- 105 may constitute a T-cell proliferative epitope in RAS. We postulate that the high load of micro-organisms that colonize the oral mucosa may initiate an immune response by the microbial HSP 65-derived peptide 95-105, stimulating the numerous Langerhans cells in the oral mucosa to activate a cross-reacting immune response to the homologous peptide 116-130 within the epithelial HSP 60, initiating the immunopathological changes that lead to RAS.
Figures


References
-
- Sircus W, Church R, Kelleher J. Recurrent aphthous ulceration of the mouth. Qly J Med. 1957;26:235–42. - PubMed
-
- Lehner T. Recurrent aphthous ulceration and autoimmunity. Lancet. 1964;2:1154–5. - PubMed
-
- Graykowski EA, Barile MF, Lee WB, Stanley HR. Recurrent aphthous stomatitis. Clinical, therapeutic, histopathologic and hypersensitivity aspects. J Am Med Assoc. 1966;196:637–44. - PubMed
-
- Donatsky O. A leukoctye migration study on the cell-mediated immunity against adult human oral mucosa and streptococcal antigens in patients with recurrent aphthous stomatitis. Arch Dermatol. 1974;109:361–3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

