Evaluation of the in vitro pyrogen test system based on proinflammatory cytokine release from human monocytes: comparison with a human whole blood culture test system and with the rabbit pyrogen test
- PMID: 11986265
- PMCID: PMC119983
- DOI: 10.1128/cdli.9.3.588-597.2002
Evaluation of the in vitro pyrogen test system based on proinflammatory cytokine release from human monocytes: comparison with a human whole blood culture test system and with the rabbit pyrogen test
Abstract
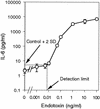
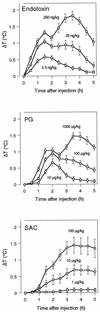
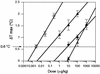
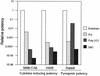
The reliability of an in vitro pyrogen test system based on proinflammatory cytokine release from human monocytic cells was assessed by comparison with a test system based on a human whole blood culture as well as with the conventional rabbit pyrogen test. The human cells used as the pyrogen indicator cells were newly selected by subcloning of a human monocytic cell line, Mono-Mac-6. The selected cells, named MM6-CA8, responded to various pyrogens, including endotoxin, peptidoglycan (PG), Staphylococcus aureus Cowan 1 (SAC), and poly(I x C), with a high sensitivity and produced proinflammatory cytokines, such as interleukin 1 (IL-1), IL-6, and tumor necrosis factor alpha. Among these cytokines, IL-6 was produced most sensitively in response to traces of the pyrogens and detected in the largest quantities in the culture medium. The cytokine-producing responses of MM6-CA8 cells correlated significantly with the responses of cultured human whole blood, which represents an ex vivo culture test system reproducing pyrogen-induced cytokine production in the human body. In terms of cytokine inducibility, the pyrogens were ranked in the order endotoxin > PG > poly (I. C) > SAC in both culture systems, a ranking which almost agreed with the ranking of their pyrogenicity as assessed by the rabbit pyrogen test. These results suggest that the in vitro responsiveness of MM6-CA8 cells to various pyrogens is highly relevant for human pyrogenic reactions. Therefore, the in vitro test system is useful and reliable for detecting the presence of materials that are pyrogenic for humans.
Figures



Similar articles
-
Soluble β-(1,3)-glucans enhance LPS-induced response in the monocyte activation test, but inhibit LPS-mediated febrile response in rabbits: Implications for pyrogenicity tests.Eur J Pharm Sci. 2016 Jan 1;81:18-26. doi: 10.1016/j.ejps.2015.09.018. Epub 2015 Sep 30. Eur J Pharm Sci. 2016. PMID: 26428698
-
A comparative study of Mono Mac 6 cells, isolated mononuclear cells and Limulus amoebocyte lysate assay in pyrogen testing.Int J Pharm. 1999 Nov 30;191(2):141-9. doi: 10.1016/s0378-5173(99)00294-x. Int J Pharm. 1999. PMID: 10564840
-
Endotoxin contamination in wound dressings made of natural biomaterials.J Biomed Mater Res B Appl Biomater. 2003 Jul 15;66(1):347-55. doi: 10.1002/jbm.b.10020. J Biomed Mater Res B Appl Biomater. 2003. PMID: 12808594
-
Material-mediated pyrogens in medical devices: Applicability of the in vitro Monocyte Activation Test.ALTEX. 2018;35(4):453-463. doi: 10.14573/altex.1709221. Epub 2018 Jun 14. ALTEX. 2018. PMID: 29901209 Review.
-
More than 70 years of pyrogen detection: Current state and future perspectives.Altern Lab Anim. 2016 Jul;44(3):239-53. doi: 10.1177/026119291604400305. Altern Lab Anim. 2016. PMID: 27494624 Review.
Cited by
-
Innate Immunity Modulating Impurities and the Immunotoxicity of Nanobiotechnology-Based Drug Products.Molecules. 2021 Dec 1;26(23):7308. doi: 10.3390/molecules26237308. Molecules. 2021. PMID: 34885886 Free PMC article. Review.
-
Homologous high-throughput expression and purification of highly conserved E coli proteins.Microb Cell Fact. 2007 Jun 6;6:18. doi: 10.1186/1475-2859-6-18. Microb Cell Fact. 2007. PMID: 17553160 Free PMC article.
-
Comparison of the interleukin-1β-inducing potency of allergenic spores from higher fungi (basidiomycetes) in a cryopreserved human whole blood system.Int Arch Allergy Immunol. 2014;163(2):154-62. doi: 10.1159/000357036. Epub 2013 Dec 17. Int Arch Allergy Immunol. 2014. PMID: 24356469 Free PMC article.
-
Comment on: Harte et al. High fat intake leads to acute postprandial exposure to circulating endotoxin in type 2 diabetic subjects. Diabetes Care 2012;35:375-382.Diabetes Care. 2013 Mar;36(3):e42. doi: 10.2337/dc12-0813. Diabetes Care. 2013. PMID: 23431104 Free PMC article. No abstract available.
-
Detection of innate immune response modulating impurities in therapeutic proteins.PLoS One. 2015 Apr 22;10(4):e0125078. doi: 10.1371/journal.pone.0125078. eCollection 2015. PLoS One. 2015. PMID: 25901912 Free PMC article.
References
-
- Abel, G., and J. K. Czop. 1992. Stimulation of human monocyte beta-glucan receptors by glucan induces production of TNF-alpha and IL-1 beta. Int. J. Immunopharmacol. 14:1363-1373. - PubMed
-
- Cohen, J., and E. Abraham. 1999. Microbiological findings and correlations with serum tumor necrosis factor-α in patients with severe sepsis and septic shock. J. Infect. Dis. 180:116-121. - PubMed
-
- Cornell, J. C. J., K. A. Smith, G. G. Cornwell, I. I. I., G. P. Burke, and O. R. Mclntyre. 1976. Systemic effects of intravenous polyriboinsinic-polyribocytidylic acid in man. J. Natl. Cancer Inst. 57:1211-1216. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

