Diarrhea-inducing activity of avian rotavirus NSP4 glycoproteins, which differ greatly from mammalian rotavirus NSP4 glycoproteins in deduced amino acid sequence in suckling mice
- PMID: 11992013
- PMCID: PMC137017
- DOI: 10.1128/jvi.76.11.5829-5834.2002
Diarrhea-inducing activity of avian rotavirus NSP4 glycoproteins, which differ greatly from mammalian rotavirus NSP4 glycoproteins in deduced amino acid sequence in suckling mice
Abstract
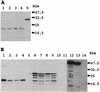
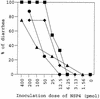
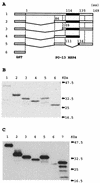
Avian rotavirus NSP4 glycoproteins expressed in Escherichia coli acted as enterotoxins in suckling mice, as did mammalian rotavirus NSP4 glycoproteins, despite great differences in the amino acid sequences. The enterotoxin domain of PO-13 NSP4 exists in amino acid residues 109 to 135, a region similar to that reported in SA11 NSP4.
Figures
References
-
- Ball, J. M., P. Tian, C. Zeng, A. P. Morris, and M. K. Estes. 1996. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science 272:101-104. - PubMed
-
- Both, G. W., A. R. Bellamy, and D. B. Mitchell. 1994. Rotavirus protein structure and function. Curr. Top. Microbiol. Immunol. 185:67-105. - PubMed
-
- Bowman, G. D., I. M. Nodelman, O. Levy, S. L. Lin, P. Tian, T. J. Zamb, S. A. Udem, B. Venkataraghavan, and C. E. Schutt. 2000. Crystal structure of the oligomerization domain of NSP4 from rotavirus reveals a core metal-binding site. J. Mol. Biol. 304:861-871. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

