Pharmacokinetics and pharmacodynamics of combination chemotherapy with paclitaxel and epirubicin in breast cancer patients
- PMID: 11994057
- PMCID: PMC1874362
- DOI: 10.1046/j.1365-2125.2002.01579.x
Pharmacokinetics and pharmacodynamics of combination chemotherapy with paclitaxel and epirubicin in breast cancer patients
Abstract
Aims: To investigate the pharmacokinetics and pharmacodynamics of epirubicin and paclitaxel in combination, as well as the effects of paclitaxel and its vehicle Cremophor EL on epirubicin metabolism.
Methods: Twenty-seven female patients with metastatic breast cancer received epirubicin 90 mg m-2 i.v. followed 15 min or 30 h later by a 3 h i.v. infusion of paclitaxel 175, 200 and 225 mg m-2. Plasma concentrations of paclitaxel, epirubicin and epirubicinol were measured and the relationship between neutropenia and drug pharmacokinetics was evaluated using a sigmoid maximum effect (Emax) model. Finally, the influence of paclitaxel and Cremophor EL on epirubicin metabolism by whole blood was examined.
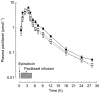
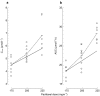
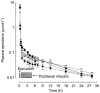
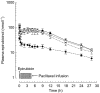
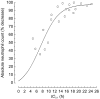
Results: An increase in epirubicinol plasma concentrations occurred after the start of the paclitaxel infusion, resulting in a significant increase in the area under the plasma concentration-time curve (AUC) of epirubicinol (+0.5 micromol l-1 h [95% CI for the difference: 0.29, 0.71],+0.66 micromol l-1 h [95% CI for the difference: 0.47, 0.85] and +0.82 micromol l-1 h [95% CI for the difference: 0.53, 1.11] at paclitaxel doses of 175, 200 and 225 mg m-2, respectively), compared with epirubicin followed by paclitaxel 30 h later (0.61+/-0.1 micromol l-1 h). A significant increase in epirubicin AUC (+0.74 micromol l-1 h [95% CI for the difference: 0.14, 1.34] and +1.09 micromol l-1 h [95% CI for the difference: 0.44, 1.74]) and decrease in drug clearance (CLTB) (-25.35 l h-1 m-2[95% CI for the difference: -50.18, -0.52] and -35.9 l h-1 m-2[95% CI for the difference -63,4,-8,36]) occurred in combination with paclitaxel 200 and 225 mg m-2 with respect to the AUC (3.16+/-0.6 micromol l-1 h) and CLTB (74.4+/-28.4 l h-1 m-2) of epirubicin followed by paclitaxel 30 h later. An Emax relationship was observed between neutropaenia and the time over which paclitaxel plasma concentrations were equal to or greater than 0.1 micromol l-1 (tC0.1). The tC0.1 value predicted to yield a 50% decrease in neutrophil count was 7.7 h. Finally, Cremophor EL markedly inhibited the metabolism of epirubicin to epirubicinol in whole blood.
Conclusions: Paclitaxel/Cremophor EL affects the disposition of epirubicinol and epirubicin. Furthermore, the slope factor of the Emax relationship between neutropenia and tC0.1 of paclitaxel suggests that the drugs might also interact at the pharmacodynamic level.
Figures

References
-
- Miller KD, Sledge GW., Jr Taxanes in the treatment of breast cancer: a prodigy comes of age. Cancer Invest. 1999;17:121–136. - PubMed
-
- Kaufman PA. Paclitaxel and anthracycline combination chemotherapy for metastatic breast cancer. Semin Oncol. 1999;26:39–46. - PubMed
-
- Gianni L, Munzone E, Capri G, et al. Paclitaxel by 3-hour infusion in combination with bolus doxorubicin in women with untreated metastatic breast cancer. High antitumor efficacy and cardiac effects in a dose-finding and sequence-finding study. J Clin Oncol. 1995;13:2688–2699. - PubMed
-
- Gehl J, Boesgaard M, Paaske T, Vittrup Jensen B, Dombernowsky P. Combined doxorubicin and paclitaxel in advanced breast cancer: effective and cardiotoxic. Ann Oncol. 1996;7:687–693. - PubMed
-
- Holmes FA, Madden T, Newman RA, et al. Sequence-dependent alteration of doxorubicin pharmacokinetics by paclitaxel in a phase I study of paclitaxel and doxorubicin in patients with metastatic breast cancer. J Clin Oncol. 1996;14:2713–2721. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

