Determinants of the cellular specificity of acetaminophen as an inhibitor of prostaglandin H(2) synthases
- PMID: 12011469
- PMCID: PMC124540
- DOI: 10.1073/pnas.102588199
Determinants of the cellular specificity of acetaminophen as an inhibitor of prostaglandin H(2) synthases
Abstract
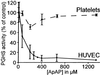
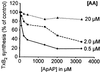
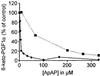
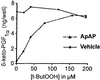
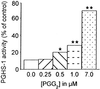
Acetaminophen has antipyretic and analgesic properties yet differs from the nonsteroidal antiinflammatory drugs and inhibitors of prostaglandin H synthase (PGHS)-2 by exhibiting little effect on platelets or inflammation. We find parallel selectivity at a cellular level; acetaminophen inhibits PGHS activity with an IC(50) of 4.3 microM in interleukin (IL)-1 alpha-stimulated human umbilical vein endothelial cells, in contrast with an IC(50) of 1,870 microM for the platelet, with 2 microM arachidonic acid as substrate. This difference is not caused by isoform selectivity, because acetaminophen inhibits purified ovine PGHS-1 and murine recombinant PGHS-2 equally. We explored the hypothesis that this difference in cellular responsiveness results from antagonism of the reductant action of acetaminophen on the PGHSs by cellular peroxides. Increasing the peroxide product of the PGHS-cyclooxygenase, prostaglandin G(2) (PGG(2)), by elevating the concentration of either enzyme or substrate reverses the inhibitory action of acetaminophen, as does the addition of PGG(2) itself. 12-Hydroperoxyeicosatetraenoic acid (0.3 microM), a major product of the platelet, completely reverses the action of acetaminophen on PGHS-1. Inhibition of PGHS activity by acetaminophen in human umbilical vein endothelial cells is abrogated by t-butyl hydroperoxide. Together these findings support the hypothesis that the clinical action of acetaminophen is mediated by inhibition of PGHS activity, and that hydroperoxide concentration contributes to its cellular selectivity.
Figures



References
-
- Pernerstorfer T, Schmid R, Bieglmayer C, Eichler H G, Kapiotis S, Jilma B. Clin Pharmacol Ther. 1999;66:51–57. - PubMed
-
- Yaffe S J. Arch Intern Med (Moscow) 1981;141:286–292. - PubMed
-
- Cooper S A. Arch Intern Med (Moscow) 1981;141:282–285. - PubMed
-
- Cooper S A. Annu Rev Pharmacol Toxicol. 1983;23:617–647. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

