Connection between elastin haploinsufficiency and increased cell proliferation in patients with supravalvular aortic stenosis and Williams-Beuren syndrome
- PMID: 12016585
- PMCID: PMC384991
- DOI: 10.1086/341035
Connection between elastin haploinsufficiency and increased cell proliferation in patients with supravalvular aortic stenosis and Williams-Beuren syndrome
Abstract
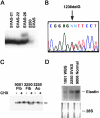
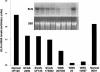
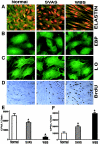
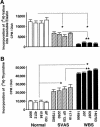
To elucidate the pathomechanism leading to obstructive vascular disease in patients with elastin deficiency, we compared both elastogenesis and proliferation rate of cultured aortic smooth-muscle cells (SMCs) and skin fibroblasts from five healthy control subjects, four patients with isolated supravalvular aortic stenosis (SVAS), and five patients with Williams-Beuren syndrome (WBS). Mutations were determined in each patient with SVAS and in each patient with WBS. Three mutations found in patients with SVAS were shown to result in null alleles. RNA blot hybridization, immunostaining, and metabolic labeling experiments demonstrated that SVAS cells and WBS cells have reduced elastin mRNA levels and that they consequently deposit low amounts of insoluble elastin. Although SVAS cells laid down approximately 50% of the elastin made by normal cells, WBS cells deposited only 15% of the elastin made by normal cells. The observed difference in elastin-gene expression was not caused by a difference in the stability of elastin mRNA in SVAS cells compared with WBS cells, but it did indicate that gene-interaction effects may contribute to the complex phenotype observed in patients with WBS. Abnormally low levels of elastin deposition in SVAS cells and in WBS cells were found to coincide with an increase in proliferation rate, which could be reversed by addition of exogenous insoluble elastin. We conclude that insoluble elastin is an important regulator of cellular proliferation. Thus, the reduced net deposition of insoluble elastin in arterial walls of patients with either SVAS or WBS leads to the increased proliferation of arterial SMCs. This results in the formation of multilayer thickening of the tunica media of large arteries and, consequently, in the development of hyperplastic intimal lesions leading to segmental arterial occlusion.
Figures

Similar articles
-
Vascular wall remodeling in patients with supravalvular aortic stenosis and Williams Beuren syndrome.J Vasc Res. 2005 May-Jun;42(3):190-201. doi: 10.1159/000085141. Epub 2005 Apr 12. J Vasc Res. 2005. PMID: 15832055
-
Genetic Diagnosis and the Severity of Cardiovascular Phenotype in Patients With Elastin Arteriopathy.Circ Genom Precis Med. 2020 Dec;13(6):e002971. doi: 10.1161/CIRCGEN.120.002971. Epub 2020 Sep 22. Circ Genom Precis Med. 2020. PMID: 32960096 Free PMC article.
-
Engineered zinc-finger proteins can compensate genetic haploinsufficiency by transcriptional activation of the wild-type allele: application to Willams-Beuren syndrome and supravalvular aortic stenosis.Hum Gene Ther. 2012 Nov;23(11):1186-99. doi: 10.1089/hum.2011.201. Hum Gene Ther. 2012. PMID: 22891920 Free PMC article.
-
[Role of elastin in the development of vascular function. Knock-out study of the elastin gene in mice].J Soc Biol. 2001;195(2):151-6. J Soc Biol. 2001. PMID: 11723827 Review. French.
-
Williams-Beuren syndrome: computed tomography imaging review.Pediatr Cardiol. 2014 Dec;35(8):1309-20. doi: 10.1007/s00246-014-0998-z. Epub 2014 Aug 20. Pediatr Cardiol. 2014. PMID: 25139247 Review.
Cited by
-
RNA delivery biomaterials for the treatment of genetic and rare diseases.Biomaterials. 2019 Oct;217:119291. doi: 10.1016/j.biomaterials.2019.119291. Epub 2019 Jun 20. Biomaterials. 2019. PMID: 31255978 Free PMC article. Review.
-
Neuraminidase-1, a subunit of the cell surface elastin receptor, desialylates and functionally inactivates adjacent receptors interacting with the mitogenic growth factors PDGF-BB and IGF-2.Am J Pathol. 2008 Oct;173(4):1042-56. doi: 10.2353/ajpath.2008.071081. Epub 2008 Sep 4. Am J Pathol. 2008. PMID: 18772331 Free PMC article.
-
Loss of Gata5 in mice leads to bicuspid aortic valve.J Clin Invest. 2011 Jul;121(7):2876-87. doi: 10.1172/JCI44555. J Clin Invest. 2011. PMID: 21633169 Free PMC article.
-
Fibulin-4 regulates expression of the tropoelastin gene and consequent elastic-fibre formation by human fibroblasts.Biochem J. 2009 Sep 14;423(1):79-89. doi: 10.1042/BJ20090993. Biochem J. 2009. PMID: 19627254 Free PMC article.
-
Modeling and rescue of the vascular phenotype of Williams-Beuren syndrome in patient induced pluripotent stem cells.Stem Cells Transl Med. 2013 Jan;2(1):2-15. doi: 10.5966/sctm.2012-0054. Epub 2012 Dec 21. Stem Cells Transl Med. 2013. PMID: 23283491 Free PMC article.
References
Electronic-Database Information
-
- Online Mendelian Inheritance in Man (OMIM), http://www.ncbi.nlm.nih.gov/omim (for SVAS [MIM 185500] and WBS [MIM 194050])
References
-
- Bashir MM, Indik Z, Yeh,H., Ornstein-Goldstein N, Rosenbloom JC, Abrams W, Fazio M, Uitto J, Rosenbloom J (1989) Characterization of the complete human elastin gene. J Biol Chem 264:8887–8891 - PubMed
-
- Beuren AJ, Apitz J, Harmjanz D (1962) Supravalvular aortic stenosis in association with mental retardation and a certain facial appearance. Circulation 26:1235–1240 - PubMed
-
- Boeckel T, Dierks A, Vergopoulos A, Bähring S, Knoblauch H, Müller-Myhsok B, Baron H, Aydin A, Bein G, Luft FC, Schuster H (1999) A new mutation in the elastin gene causing supravalvular aortic stenosis. Am J Cardiol 83:1141–1143 - PubMed
-
- Conway EE Jr, Noonan J, Marion RW, Steeg CN (1990) Myocardial infarction leading to sudden death in the Williams syndrome: report of three cases. J Pediatr 117:593–595 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

