Molecular dissection of hippocampal theta-burst pairing potentiation
- PMID: 12032353
- PMCID: PMC124338
- DOI: 10.1073/pnas.092157999
Molecular dissection of hippocampal theta-burst pairing potentiation
Abstract
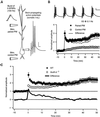
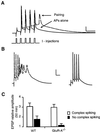
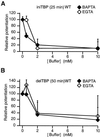
Long-term potentiation (LTP) of synaptic efficacy in the hippocampus is frequently induced by tetanic stimulation of presynaptic afferents or by pairing low frequency stimulation with postsynaptic depolarization. Adult (P42) GluR-A(-/-) mice largely lack these forms of LTP. LTP in wt mice can also be induced by coincident pre- and postsynaptic action potentials, where an initial rapid component is expressed but a substantial fraction of the potentiation develops with a delayed time course. We report here that this stimulation protocol, delivered at theta frequency (5 Hz), induces LTP in GluR-A(-/-) mice in which the initial component is substantially reduced. The remaining GluR-A independent component differs from the initial component in that its expression develops over time after induction and its induction is differentially dependent on postsynaptic intracellular Ca(2+) buffering. Thus, in adult mice, theta-burst pairing evokes two forms of synaptic potentiation that are induced simultaneously but whose expression levels vary inversely with time. The two components of synaptic potentiation could be relevant for different forms of information storage that are dependent on hippocampal synaptic transmission such as spatial reference and working memory.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

