Spatiotemporal features of early neuronogenesis differ in wild-type and albino mouse retina
- PMID: 12040030
- PMCID: PMC4127325
- DOI: 10.1523/JNEUROSCI.22-11-04249.2002
Spatiotemporal features of early neuronogenesis differ in wild-type and albino mouse retina
Abstract
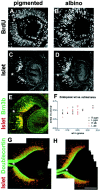
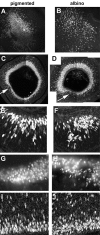
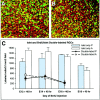
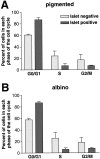
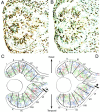
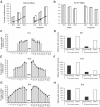
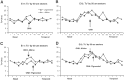
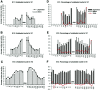
In albino mammals, lack of pigment in the retinal pigment epithelium is associated with retinal defects, including poor visual acuity from a photoreceptor deficit in the central retina and poor depth perception from a decrease in ipsilaterally projecting retinal fibers. Possible contributors to these abnormalities are reported delays in neuronogenesis (Ilia and Jeffery, 1996) and retinal maturation (Webster and Rowe, 1991). To further determine possible perturbations in neuronogenesis and/or differentiation, we used cell-specific markers and refined birth dating methods to examine these events during retinal ganglion cell (RGC) genesis in albino and pigmented mice from embryonic day 11 (E11) to E18. Our data indicate that relative to pigmented mice, more ganglion cells are born in the early stages of neuronogenesis in the albino retina, although the initiation of RGC genesis in the albino is unchanged. The cellular organization of the albino retina is perturbed as early as E12. In addition, cell cycle kinetics and output along the nasotemporal axis differ in retinas of albino and pigmented mice, both absolutely, with the temporal aspect of the retina expanded in albino, and relative to the position of the optic nerve head. Finally, blocking melanin synthesis in pigmented eyecups in culture leads to an increase in RGC differentiation, consistent with a role for melanin formation in regulating RGC neuronogenesis. These results point to spatiotemporal defects in neuronal production in the albino retina, which could perturb expression of genes that specify cell fate, number, and/or projection phenotype.
Figures

Similar articles
-
Delayed neurogenesis leads to altered specification of ventrotemporal retinal ganglion cells in albino mice.Neural Dev. 2014 May 18;9:11. doi: 10.1186/1749-8104-9-11. Neural Dev. 2014. PMID: 24885435 Free PMC article.
-
Retinal decussation patterns in pigmented and albino ferrets.Neuroscience. 1987 Feb;20(2):519-35. doi: 10.1016/0306-4522(87)90108-4. Neuroscience. 1987. PMID: 3587608
-
L-Dopa and the albino riddle: content of L-Dopa in the developing retina of pigmented and albino mice.PLoS One. 2013;8(3):e57184. doi: 10.1371/journal.pone.0057184. Epub 2013 Mar 19. PLoS One. 2013. PMID: 23526936 Free PMC article.
-
New animal models to study the role of tyrosinase in normal retinal development.Front Biosci. 2006 Jan 1;11:743-52. doi: 10.2741/1832. Front Biosci. 2006. PMID: 16146766 Review.
-
Methods to Identify Rat and Mouse Retinal Ganglion Cells in Retinal Flat-Mounts.Methods Mol Biol. 2023;2708:175-194. doi: 10.1007/978-1-0716-3409-7_18. Methods Mol Biol. 2023. PMID: 37558971 Review.
Cited by
-
The genetic control of avascular area in mouse oxygen-induced retinopathy.Mol Vis. 2012;18:377-89. Epub 2012 Feb 8. Mol Vis. 2012. PMID: 22355249 Free PMC article.
-
Retinal projections to the subcortical visual system in congenic albino and pigmented rats.Neuroscience. 2006 Dec;143(3):895-904. doi: 10.1016/j.neuroscience.2006.08.016. Epub 2006 Sep 22. Neuroscience. 2006. PMID: 16996223 Free PMC article.
-
Conversations with Ray Guillery on albinism: linking Siamese cat visual pathway connectivity to mouse retinal development.Eur J Neurosci. 2019 Apr;49(7):913-927. doi: 10.1111/ejn.14396. Epub 2019 Apr 23. Eur J Neurosci. 2019. PMID: 30801828 Free PMC article. Review.
-
The Ciliary Margin Zone of the Mammalian Retina Generates Retinal Ganglion Cells.Cell Rep. 2016 Dec 20;17(12):3153-3164. doi: 10.1016/j.celrep.2016.11.016. Cell Rep. 2016. PMID: 28009286 Free PMC article.
-
Investigation of Frizzled-5 during embryonic neural development in mouse.Dev Dyn. 2008 Jun;237(6):1614-26. doi: 10.1002/dvdy.21565. Dev Dyn. 2008. PMID: 18489003 Free PMC article.
References
-
- Alexiades M, Cepko C. Quantitative analysis of proliferation and cell cycle length during development of the rat retina. Dev Dyn. 1996;205:293–307. - PubMed
-
- Beisker W, Dolbeare F, Gray J. An improved immunocytochemical procedure for high-sensitivity detection of incorporated bromodeoxyuridine. Cytometry. 1987;8:235–239. - PubMed
-
- Brown A, Yates P, Burrola P, Ortuño D, Vaidya A, Jessell T, Pfaff S, O'Leary D, Lemke G. Topographic mapping from the retina to the midbrain is controlled by relative but not absolute levels of EphA receptor signaling. Cell. 2000;102:77–88. - PubMed
-
- Carter-Dawson L, LaVail M. Rods and cones in the mouse retina. II. Autoradiographic analysis of cell generation using tritiated thymidine. J Comp Neurol. 1979;188:263–272. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
