G-protein-gated potassium channels containing Kir3.2 and Kir3.3 subunits mediate the acute inhibitory effects of opioids on locus ceruleus neurons
- PMID: 12040038
- PMCID: PMC6758804
- DOI: 10.1523/JNEUROSCI.22-11-04328.2002
G-protein-gated potassium channels containing Kir3.2 and Kir3.3 subunits mediate the acute inhibitory effects of opioids on locus ceruleus neurons
Abstract
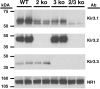
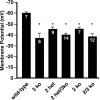
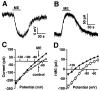
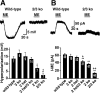
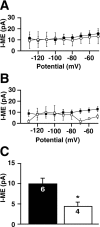
Acute opioid administration causes hyperpolarization of locus ceruleus (LC) neurons. A G-protein-gated, inwardly rectifying potassium (GIRK/K(G)) conductance and a cAMP-dependent cation conductance have both been implicated in this effect; the relative contribution of each conductance remains controversial. Here, the contribution of K(G) channels to the inhibitory effects of opioids on LC neurons was examined using mice that lack the K(G) channel subunits Kir3.2 and Kir3.3. Resting membrane potentials of LC neurons in brain slices from Kir3.2 knock-out, Kir3.3 knock-out, and Kir3.2/3.3 double knock-out mice were depolarized by 15-20 mV relative to LC neurons from wild-type mice. [Met](5)enkephalin-induced hyperpolarization and whole-cell current were reduced by 40% in LC neurons from Kir3.2 knock-out mice and by 80% in neurons from Kir3.2/3.3 double knock-out mice. The small opioid-sensitive current observed in LC neurons from Kir3.2/3.3 double knock-out mice was virtually eliminated with the nonselective potassium channel blockers barium and cesium. We conclude that the acute opioid inhibition of LC neurons is mediated primarily by the activation of G-protein-gated potassium channels and that the cAMP-dependent cation conductance does not contribute significantly to this effect.
Figures

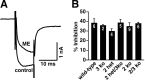
Similar articles
-
Contribution of Kir3.1, Kir3.2A and Kir3.2C subunits to native G protein-gated inwardly rectifying potassium currents in cultured hippocampal neurons.Eur J Neurosci. 2003 Oct;18(8):2110-8. doi: 10.1046/j.1460-9568.2003.02933.x. Eur J Neurosci. 2003. PMID: 14622172
-
Pre- and postsynaptic regulation of locus coeruleus neurons after chronic morphine treatment: a study of GIRK-knockout mice.Eur J Neurosci. 2008 Aug;28(3):618-24. doi: 10.1111/j.1460-9568.2008.06348.x. Eur J Neurosci. 2008. PMID: 18702733 Free PMC article.
-
Pore mutation in a G-protein-gated inwardly rectifying K+ channel subunit causes loss of K+-dependent inhibition in weaver hippocampus.J Neurosci. 1998 Jun 1;18(11):4001-7. doi: 10.1523/JNEUROSCI.18-11-04001.1998. J Neurosci. 1998. PMID: 9592081 Free PMC article.
-
Mechanisms of opioid actions on neurons of the locus coeruleus.Prog Brain Res. 1991;88:197-205. doi: 10.1016/s0079-6123(08)63809-1. Prog Brain Res. 1991. PMID: 1667545 Review.
-
The role of G proteins in assembly and function of Kir3 inwardly rectifying potassium channels.Channels (Austin). 2010 Sep-Oct;4(5):411-21. doi: 10.4161/chan.4.5.13327. Epub 2010 Sep 1. Channels (Austin). 2010. PMID: 20855978 Free PMC article. Review.
Cited by
-
Sex differences in GABA(B)R-GIRK signaling in layer 5/6 pyramidal neurons of the mouse prelimbic cortex.Neuropharmacology. 2015 Aug;95:353-60. doi: 10.1016/j.neuropharm.2015.03.029. Epub 2015 Apr 2. Neuropharmacology. 2015. PMID: 25843643 Free PMC article.
-
The K(+) channel GIRK2 is both necessary and sufficient for peripheral opioid-mediated analgesia.EMBO Mol Med. 2013 Aug;5(8):1263-77. doi: 10.1002/emmm.201201980. Epub 2013 Jul 1. EMBO Mol Med. 2013. PMID: 23818182 Free PMC article.
-
Compartment-dependent colocalization of Kir3.2-containing K+ channels and GABAB receptors in hippocampal pyramidal cells.J Neurosci. 2006 Apr 19;26(16):4289-97. doi: 10.1523/JNEUROSCI.4178-05.2006. J Neurosci. 2006. PMID: 16624949 Free PMC article.
-
G protein {beta}{gamma} gating confers volatile anesthetic inhibition to Kir3 channels.J Biol Chem. 2010 Dec 31;285(53):41290-9. doi: 10.1074/jbc.M110.178541. Epub 2010 Nov 2. J Biol Chem. 2010. PMID: 21044958 Free PMC article.
-
Activation of Astrocytic μ-opioid Receptor Elicits Fast Glutamate Release Through TREK-1-Containing K2P Channel in Hippocampal Astrocytes.Front Cell Neurosci. 2018 Sep 27;12:319. doi: 10.3389/fncel.2018.00319. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30319359 Free PMC article.
References
-
- Aghajanian G, Wang Y-Y. Pertussis toxin blocks the outward currents evoked by opiate and alpha(2)-agonists in locus coeruleus neurons. Brain Res. 1986;371:390–394. - PubMed
-
- Alreja M, Aghajanian G. QX-314 blocks the potassium but not the sodium-dependent component of the opiate response in locus coeruleus neurons. Brain Res. 1994;639:320–324. - PubMed
-
- Alreja M, Aghajanian G. Use of the whole-cell patch-clamp method in studies on the role of cAMP in regulating the spontaneous firing of locus coeruleus neurons. J Neurosci Methods. 1995;59:67–75. - PubMed
-
- Barry PH. JPCalc, a software package for calculating liquid junction potential corrections in patch-clamp, intracellular, epithelial and bilayer measurements and for correcting junction potential measurements. J Neurosci Methods. 1994;51:107–116. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
