High-molecular-weight protein hydrodynamics studied with a long-lifetime metal-ligand complex
- PMID: 12044900
- PMCID: PMC6800114
- DOI: 10.1016/s0167-4838(02)00281-9
High-molecular-weight protein hydrodynamics studied with a long-lifetime metal-ligand complex
Erratum in
- Biochim Biophys Acta 2002 Jul 29;1598(1-2):196
Abstract
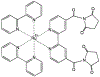
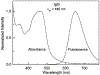
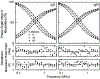
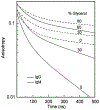
[Ru(2,2'-bipyridine)(2)(4,4'-dicarboxy-2,2'-bipyridine)](2+) (RuBDc) is a very photostable probe that possesses favorable photophysical properties including long lifetime, high quantum yield, large Stokes' shift, and highly polarized emission. In the present study, we demonstrated the usefulness of this probe for monitoring the rotational diffusion of high-molecular-weight (MW) proteins. Using frequency-domain fluorometry with a high-intensity, blue light-emitting diode (LED) as the modulated light source, we compared the intensity and anisotropy decays of RuBDc conjugated to immunoglobulin G (IgG) and immunoglobulin M (IgM), which show a six-fold difference in MW We obtained slightly longer lifetimes for IgM (<tau>=428 ns in buffer) than IgG (<tau>=422 ns in buffer) in the absence and presence of glycerol, suggesting somewhat more efficient shielding of RuBDc from water in IgM than in IgG. The anisotropy decay data showed longer rotational correlation times for IgM (1623 and 65.7 ns in buffer) as compared to IgG (264 and 42.5 ns in buffer). Importantly, the ratio of the long rotational correlation times of IgM to IgG in buffer was 6.2, which is very close to that of MW of IgM to IgG (6.0). The shorter correlation times are most likely to be associated with domain motions within the proteins. The anisotropy decays reflect both the molecular size and shape of the immunoglobulins, as well as the viscosity. These results show that RuBDc can have numerous applications in studies of high-MW protein hydrodynamics and in fluorescence polarization immunoassays (FPI) of high-MW analytes.
Figures

Similar articles
-
Dynamics of bacteriophage R17 probed with a long-lifetime Ru(II) metal-ligand complex.J Fluoresc. 2010 May;20(3):713-8. doi: 10.1007/s10895-010-0612-6. Epub 2010 Feb 27. J Fluoresc. 2010. PMID: 20195712
-
Fluorescence polarization immunoassay of a high-molecular-weight antigen based on a long-lifetime Ru-ligand complex.Anal Biochem. 1995 May 1;227(1):140-7. doi: 10.1006/abio.1995.1263. Anal Biochem. 1995. PMID: 7668374 Free PMC article.
-
Long-lifetime metal-ligand pH probe.Anal Biochem. 1997 May 1;247(2):216-22. doi: 10.1006/abio.1997.2057. Anal Biochem. 1997. PMID: 9177680 Free PMC article.
-
Dynamics of supercoiled and relaxed pTZ18U plasmids probed with a long-lifetime metal-ligand complex.J Biochem Mol Biol. 2002 Jul 31;35(4):389-94. doi: 10.5483/bmbrep.2002.35.4.389. J Biochem Mol Biol. 2002. PMID: 12296998 Free PMC article.
-
Dynamics of supercoiled and linear pTZ18U plasmids observed with a long-lifetime metal-ligand complex.Biopolymers. 2002;67(2):121-8. doi: 10.1002/bip.10057. Biopolymers. 2002. PMID: 12073934
Cited by
-
Dynamics of bacteriophage R17 probed with a long-lifetime Ru(II) metal-ligand complex.J Fluoresc. 2010 May;20(3):713-8. doi: 10.1007/s10895-010-0612-6. Epub 2010 Feb 27. J Fluoresc. 2010. PMID: 20195712
-
Fluorophore Interactions with the Surface Modes and Internal Modes of a Photonic Crystal.Opt Mater (Amst). 2024 Jan;147:114718. doi: 10.1016/j.optmat.2023.114718. Epub 2023 Dec 15. Opt Mater (Amst). 2024. PMID: 38283740 Free PMC article.
References
-
- Badea MG, Brand L, Methods Enzymol. 61 (1979) 378–425. - PubMed
-
- DeGraff BA, Demas JN, J. Phys. Chem. 98 (1994) 12478–12480.
-
- Lakowicz JR, Gryczynski I, Piszczek G, Tolosa L, Nair R, Johnson LM, Nowaczyk K, Methods Enzymol. 323 (2000) 473–509. - PubMed
-
- Terpetschnig E, Szmacinski H, Lakowicz JR, Methods Enzymol. 278 (1997) 295–321. - PubMed
-
- Castellano FN, Dattelbaum JD, Lakowicz JR, Anal. Biochem. 255 (1998) 165–170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

