Caveolin-1/3 double-knockout mice are viable, but lack both muscle and non-muscle caveolae, and develop a severe cardiomyopathic phenotype
- PMID: 12057923
- PMCID: PMC1850810
- DOI: 10.1016/S0002-9440(10)61168-6
Caveolin-1/3 double-knockout mice are viable, but lack both muscle and non-muscle caveolae, and develop a severe cardiomyopathic phenotype
Abstract
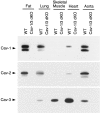
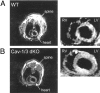
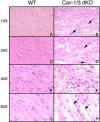
The caveolin gene family consists of caveolins 1, 2, and 3. Caveolins 1 and 2 are co-expressed in many cell types, such as endothelial cells, fibroblasts, smooth muscle cells and adipocytes, where they form a heteroligomeric complex. In contrast, the expression of caveolin-3 is muscle-specific. Thus, the expression of caveolin-1 is required for caveolae formation in non-muscle cells, while the expression of caveolin-3 drives caveolae formation in striated muscle cell types (cardiac and skeletal). To create a truly caveolae-deficient mouse, we interbred Cav-1 null mice and Cav-3 null mice to generate Cav-1/Cav-3 double-knockout (Cav-1/3 dKO) mice. Here, we report that Cav-1/3 dKO mice are viable and fertile, despite the fact that they lack morphologically identifiable caveolae in endothelia, adipocytes, smooth muscle cells, skeletal muscle fibers, and cardiac myocytes. We also show that these mice are deficient in all three caveolin gene products, as caveolin-2 is unstable in the absence of caveolin-1. Interestingly, Cav-1/3 dKO mice develop a severe cardiomyopathy. At 2 months of age, analysis of Cav-1/3 dKO hearts via gated magnetic resonance imaging reveals a dramatic increase in left ventricular wall thickness, as compared with Cav-1-KO, Cav-3 KO, and wild-type mice. Further functional analysis of Cav-1/3 dKO hearts via transthoracic echocardiography demonstrates hypertrophy and dilation of the left ventricle, with a significant decrease in fractional shortening. As predicted, Northern analysis of RNA derived from the left ventricle of Cav-1/3 dKO mice shows a dramatic up-regulation of the atrial natriuretic factor message, a well-established biochemical marker of cardiac hypertrophy. Finally, histological analysis of Cav-1/3 dKO hearts reveals hypertrophy, disorganization, and degeneration of the cardiac myocytes, as well as chronic interstitial fibrosis and inflammation. Thus, dual ablation of both Cav-1 and Cav-3 genes in mice leads to a pleiotropic defect in caveolae formation and severe cardiomyopathy.
Figures



Similar articles
-
Caveolin-3 knock-out mice develop a progressive cardiomyopathy and show hyperactivation of the p42/44 MAPK cascade.J Biol Chem. 2002 Oct 11;277(41):38988-97. doi: 10.1074/jbc.M205511200. Epub 2002 Jul 23. J Biol Chem. 2002. PMID: 12138167
-
Caveolin-1 null mice develop cardiac hypertrophy with hyperactivation of p42/44 MAP kinase in cardiac fibroblasts.Am J Physiol Cell Physiol. 2003 Feb;284(2):C457-74. doi: 10.1152/ajpcell.00380.2002. Epub 2002 Oct 9. Am J Physiol Cell Physiol. 2003. PMID: 12388077
-
Caveolin-1 null (-/-) mice show dramatic reductions in life span.Biochemistry. 2003 Dec 30;42(51):15124-31. doi: 10.1021/bi0356348. Biochemistry. 2003. PMID: 14690422
-
The biology of caveolae: lessons from caveolin knockout mice and implications for human disease.Mol Interv. 2003 Dec;3(8):445-64. doi: 10.1124/mi.3.8.445. Mol Interv. 2003. PMID: 14993453 Review.
-
The Caveolin genes: from cell biology to medicine.Ann Med. 2004;36(8):584-95. doi: 10.1080/07853890410018899. Ann Med. 2004. PMID: 15768830 Review.
Cited by
-
Sink or swim: lipid rafts in parasite pathogenesis.Trends Parasitol. 2012 Oct;28(10):417-26. doi: 10.1016/j.pt.2012.07.002. Epub 2012 Aug 18. Trends Parasitol. 2012. PMID: 22906512 Free PMC article. Review.
-
Deciphering Non-coding RNAs in Cardiovascular Health and Disease.Front Cardiovasc Med. 2018 Jul 2;5:73. doi: 10.3389/fcvm.2018.00073. eCollection 2018. Front Cardiovasc Med. 2018. PMID: 30013975 Free PMC article. Review.
-
Ascidian caveolin induces membrane curvature and protects tissue integrity and morphology during embryogenesis.FASEB J. 2020 Jan;34(1):1345-1361. doi: 10.1096/fj.201901281R. Epub 2019 Nov 30. FASEB J. 2020. PMID: 31914618 Free PMC article.
-
Affinity-Guided Design of Caveolin-1 Ligands for Deoligomerization.J Med Chem. 2016 Apr 28;59(8):4019-25. doi: 10.1021/acs.jmedchem.5b01536. Epub 2016 Apr 11. J Med Chem. 2016. PMID: 27010220 Free PMC article.
-
Myocyte membrane and microdomain modifications in diabetes: determinants of ischemic tolerance and cardioprotection.Cardiovasc Diabetol. 2017 Dec 4;16(1):155. doi: 10.1186/s12933-017-0638-z. Cardiovasc Diabetol. 2017. PMID: 29202762 Free PMC article. Review.
References
-
- Song KS, Scherer PE, Tang Z, Okamoto T, Li S, Chafel M, Chu C, Kohtz DS, Lisanti MP: Expression of caveolin-3 in skeletal, cardiac, and smooth muscle cells: caveolin-3 is a component of the sarcolemma and co-fractionates with dystrophin and dystrophin-associated glycoproteins J Biol Chem 1996, 271:15160-15165 - PubMed
-
- Galbiati F, Razani B, Lisanti MP: Role of caveolae and caveolin-3 in muscular dystrophy. Trends in Molecular Medicine 2001, 7:435-441 - PubMed
-
- Galbiati F, Razani B, Lisanti MP: Emerging themes in lipid rafts and caveolae. Cell 2001, 106:403-411 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

