The biology of the combretastatins as tumour vascular targeting agents
- PMID: 12059907
- PMCID: PMC2517662
- DOI: 10.1046/j.1365-2613.2002.00211.x
The biology of the combretastatins as tumour vascular targeting agents
Abstract
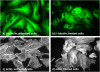
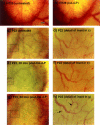
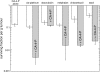
The tumour vasculature is an attractive target for therapy. Combretastatin A-4 (CA-4) and A-1 (CA-1) are tubulin binding agents, structurally related to colchicine, which induce vascular-mediated tumour necrosis in animal models. CA-1 and CA-4 were isolated from the African bush willow, Combretum caffrum, and several synthetic analogues are also now available, such as the Aventis Pharma compound, AVE8062. More soluble, phosphated, forms of CA-4 (CA-4-P) and CA-1 (CA-1-P) are commonly used for in vitro and in vivo studies. These are cleaved to the natural forms by endogenous phosphatases and are taken up into cells. The lead compound, CA-4-P, is currently in clinical trial as a tumour vascular targeting agent. In animal models, CA-4-P causes a prolonged and extensive shut-down of blood flow in established tumour blood vessels, with much less effect in normal tissues. This paper reviews the current understanding of the mechanism of action of the combretastatins and their therapeutic potential.
Figures




Similar articles
-
Disrupting tumour blood vessels.Nat Rev Cancer. 2005 Jun;5(6):423-35. doi: 10.1038/nrc1628. Nat Rev Cancer. 2005. PMID: 15928673 Review.
-
Biological potential and structure-activity relationships of most recently developed vascular disrupting agents: an overview of new derivatives of natural combretastatin a-4.Curr Med Chem. 2011;18(20):3035-81. doi: 10.2174/092986711796391642. Curr Med Chem. 2011. PMID: 21651481 Review.
-
Blocking Blood Flow to Solid Tumors by Destabilizing Tubulin: An Approach to Targeting Tumor Growth.J Med Chem. 2016 Oct 13;59(19):8685-8711. doi: 10.1021/acs.jmedchem.6b00463. Epub 2016 Jun 27. J Med Chem. 2016. PMID: 27348355
-
Mechanisms associated with tumor vascular shut-down induced by combretastatin A-4 phosphate: intravital microscopy and measurement of vascular permeability.Cancer Res. 2001 Sep 1;61(17):6413-22. Cancer Res. 2001. PMID: 11522635
-
Targeting tumour vasculature: the development of combretastatin A4.Lancet Oncol. 2001 Feb;2(2):82-7. doi: 10.1016/S1470-2045(00)00224-2. Lancet Oncol. 2001. PMID: 11905799 Review.
Cited by
-
Combretastatin A4 phosphate treatment induces vasculogenic mimicry formation of W256 breast carcinoma tumor in vitro and in vivo.Tumour Biol. 2015 Nov;36(11):8499-510. doi: 10.1007/s13277-015-3508-x. Epub 2015 May 31. Tumour Biol. 2015. PMID: 26026583
-
Site-Specific Drug-Releasing Polypeptide Nanocarriers Based on Dual-pH Response for Enhanced Therapeutic Efficacy against Drug-Resistant Tumors.Theranostics. 2015 Apr 28;5(8):890-904. doi: 10.7150/thno.11821. eCollection 2015. Theranostics. 2015. PMID: 26000060 Free PMC article.
-
The use of African medicinal plants in cancer management.Front Pharmacol. 2023 Feb 14;14:1122388. doi: 10.3389/fphar.2023.1122388. eCollection 2023. Front Pharmacol. 2023. PMID: 36865913 Free PMC article. Review.
-
Synthesis and biological evaluation of structurally diverse α-conformationally restricted chalcones and related analogues.Medchemcomm. 2019 Jun 4;10(8):1445-1456. doi: 10.1039/c9md00127a. eCollection 2019 Aug 1. Medchemcomm. 2019. PMID: 31534659 Free PMC article.
-
Necrosis targeted radiotherapy with iodine-131-labeled hypericin to improve anticancer efficacy of vascular disrupting treatment in rabbit VX2 tumor models.Oncotarget. 2015 Jun 10;6(16):14247-59. doi: 10.18632/oncotarget.3679. Oncotarget. 2015. PMID: 26036625 Free PMC article.
References
-
- Aleksandrzak K, McGown AT, Hadfield JA. Antimitotic activity of diaryl compounds with structures resembling combretastatin A-4. Anticancer Drugs. 1998;9 - PubMed
-
- Algire GH, Legallais FY, Anderson BF. Vascular reactions of normal and malignant tissues in vivo. VI. The role of hypotension in the action of components of podophyllotoxin on transplanted sarcomas. J. Natl. Cancer Inst. 1954;14:879–887. - PubMed
-
- Alon T, Hemo I, Itin A, Pe'er J, Stone J, Keshet E. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nat. Med. 1995;1:1024–1028. - PubMed
-
- Arap W, Pasqualini R, Ruoslahti E. Cancer treatment by targeted drug delivery to tumor vasculature in a mouse model. Science. 1998;279:377–380. - PubMed
-
- Baguley BC, Holdaway KM, Thomsen LL, Zhuang L, Zwi LJ. Inhibition of growth of colon 38 adenocarcinoma by vinblastine and colchicine: evidence for a vascular mechanism. Eur. J. Cancer. 1991;27:482–487. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

