Persistence of protective immunity to malaria induced by DNA priming and poxvirus boosting: characterization of effector and memory CD8(+)-T-cell populations
- PMID: 12065488
- PMCID: PMC128102
- DOI: 10.1128/IAI.70.7.3493-3499.2002
Persistence of protective immunity to malaria induced by DNA priming and poxvirus boosting: characterization of effector and memory CD8(+)-T-cell populations
Abstract
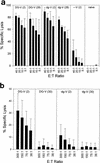
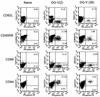
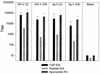
The persistence of immunity to malaria induced in mice by a heterologous DNA priming and poxvirus boosting regimen was characterized. Mice were immunized by priming with DNA vaccine plasmids encoding the Plasmodium yoelii circumsporozoite protein (PyCSP) and murine granulocyte-macrophage colony-stimulating factor and boosting with recombinant vaccinia encoding PyCSP. BALB/c mice immunized with either high-dose (100 microg of p PyCSP plus 30 microg of pGM-CSF) or low-dose (1 microg of p PyCSP plus 1 microg of pGM-CSF DNA) priming were protected against challenge with 50 P. yoelii sporozoites. Protection 2 weeks after immunization was 70 to 100%, persisted at this level for at least 20 weeks, and declined to 30 to 40% by 28 weeks. Eight of eight mice protected at 20 weeks were still protected when rechallenged at 40 weeks. The antigen (Ag)-specific effector CD8(+)-T-cell population present 2 weeks after boosting had ex vivo Ag-specific cytolytic activity, expressed both gamma interferon (IFN-gamma) and tumor necrosis factor alpha, and constituted 12 to 20% of splenic CD8(+) T cells. In contrast, the memory CD8(+)-Ag-specific-cell population at 28 weeks lacked cytolytic activity and constituted only 6% of splenic CD8(+) T cells, but at the single-cell level it produced significantly higher levels of IFN-gamma than the effectors. High levels of Ag- or parasite-specific antibodies present 2 weeks after boosting had declined three- to sevenfold by 28 weeks. Low-dose priming was similarly immunogenic and as protective as high-dose priming against a 50-, but not a 250-, sporozoite challenge. These results demonstrate that a heterologous priming and boosting vaccination can provide lasting protection against malaria in this model system.
Figures

Similar articles
-
Improving protective immunity induced by DNA-based immunization: priming with antigen and GM-CSF-encoding plasmid DNA and boosting with antigen-expressing recombinant poxvirus.J Immunol. 2000 Jun 1;164(11):5905-12. doi: 10.4049/jimmunol.164.11.5905. J Immunol. 2000. PMID: 10820272
-
Protection of rhesus macaques against lethal Plasmodium knowlesi malaria by a heterologous DNA priming and poxvirus boosting immunization regimen.Infect Immun. 2002 Aug;70(8):4329-35. doi: 10.1128/IAI.70.8.4329-4335.2002. Infect Immun. 2002. PMID: 12117942 Free PMC article.
-
Boosting with recombinant vaccinia increases immunogenicity and protective efficacy of malaria DNA vaccine.Proc Natl Acad Sci U S A. 1998 Jun 23;95(13):7648-53. doi: 10.1073/pnas.95.13.7648. Proc Natl Acad Sci U S A. 1998. PMID: 9636204 Free PMC article.
-
Immunological responses of neonates and infants to DNA vaccines.Methods Mol Med. 2006;127:239-51. doi: 10.1385/1-59745-168-1:239. Methods Mol Med. 2006. PMID: 16988458 Review.
-
The development of a multivalent DNA vaccine for malaria.Springer Semin Immunopathol. 1997;19(2):147-59. doi: 10.1007/BF00870265. Springer Semin Immunopathol. 1997. PMID: 9406343 Review. No abstract available.
Cited by
-
Adjuvant-like effect of vaccinia virus 14K protein: a case study with malaria vaccine based on the circumsporozoite protein.J Immunol. 2012 Jun 15;188(12):6407-17. doi: 10.4049/jimmunol.1102492. Epub 2012 May 21. J Immunol. 2012. PMID: 22615208 Free PMC article.
-
Heterologous vaccination targeting prostatic acid phosphatase (PAP) using DNA and Listeria vaccines elicits superior anti-tumor immunity dependent on CD4+ T cells elicited by DNA priming.Oncoimmunology. 2018 May 7;7(8):e1456603. doi: 10.1080/2162402X.2018.1456603. eCollection 2018. Oncoimmunology. 2018. PMID: 30221049 Free PMC article.
-
Potency, efficacy and durability of DNA/DNA, DNA/protein and protein/protein based vaccination using gp63 against Leishmania donovani in BALB/c mice.PLoS One. 2011 Feb 2;6(2):e14644. doi: 10.1371/journal.pone.0014644. PLoS One. 2011. PMID: 21311597 Free PMC article.
-
Genetic vaccination approaches against malaria based on the circumsporozoite protein.Wien Klin Wochenschr. 2006;118(19-20 Suppl 3):9-17. doi: 10.1007/s00508-006-0676-0. Wien Klin Wochenschr. 2006. PMID: 17131235 Review.
-
Adenovirus 5-vectored P. falciparum vaccine expressing CSP and AMA1. Part A: safety and immunogenicity in seronegative adults.PLoS One. 2011;6(10):e24586. doi: 10.1371/journal.pone.0024586. Epub 2011 Oct 7. PLoS One. 2011. PMID: 22003383 Free PMC article. Clinical Trial.
References
-
- Amara, R. R., F. Villinger, J. D. Altman, S. L. Lydy, S. P. O'Neil, S. I. Staprans, D. C. Montefiori, Y. Xu, J. G. Herndon, L. S. Wyatt, M. A. Candido, N. L. Kozyr, P. L. Earl, J. M. Smith, H.-L. Ma, B. D. Grimm, M. L. Hulsey, J. Miller, H. M. McClure, J. M. McNicholl, B. Moss, and H. L. Robinson. 2001. Control of a mucosal challenge and prevention of AIDS by a multiprotein DNA/MVA vaccine. Science 292:69-74. - PubMed
-
- Bachmann, M. F., M. Barner, A. Viola, and M. Kopf. 1999. Distinct kinetics of cytokine production and cytolysis in effector and memory T cells after viral infection. Eur. J. Immunol. 29:291-299. - PubMed
-
- Charoenvit, Y., S. Mellouk, C. Cole, R. Bechara, M. F. Leef, M. Sedegah, L. F. Yuan, F. A. Robey, R. L. Beaudoin, and S. L. Hoffman. 1991. Monoclonal, but not polyclonal antibodies protect against Plasmodium yoeli sporozoites. J. Immunol. 146:1020-1025. - PubMed
-
- Doolan, D. L., and S. L. Hoffman. 1999. IL-12 and NK cells are required for antigen-specific adaptive immunity against malaria initiated by CD8+ T cells in the Plasmodium yoelii model. J. Immunol. 163:884-892. - PubMed
-
- Doolan, D. L., and S. L. Hoffman. 2000. The complexity of protective immunity against liver-stage malaria. J. Immunol. 165:1453-1462. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

