Different regional effects of voluntary exercise on the mechanical and electrical properties of rat ventricular myocytes
- PMID: 12068046
- PMCID: PMC2290358
- DOI: 10.1113/jphysiol.2001.013415
Different regional effects of voluntary exercise on the mechanical and electrical properties of rat ventricular myocytes
Abstract
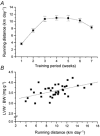
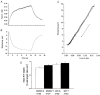
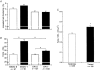
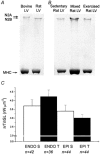
Short-term (6 weeks) voluntary wheel running exercise in young female rats that were in an active growth phase resulted in whole-heart hypertrophy and myocyte concentric hypertrophy, when compared to sedentary controls. The cross-sectional area of ventricular myocytes from trained rats was significantly greater than for those isolated from sedentary rats, with the greatest change in morphology seen in sub-endocardial cells. There was no statistically significant effect of training on cell shortening in the absence of external mechanical loading, in [Ca2+](i) transients, or in myofilament Ca2+ sensitivity (assessed during re-lengthening following tetanic stimulation). Under the external mechanical load of carbon fibres, absolute force developed in myocytes from trained rats was significantly greater than in those from sedentary rats. This suggests that increased myocyte cross-sectional area is a major contractile adaptation to exercise in this model. Training did not alter the passive mechanical properties of myocytes or the relative distribution of titin isomers, which was exclusively of the short, N2B form. However, training did increase the steepness of the active tension-sarcomere length relationship, suggesting an exercise-induced modulation of the Frank-Starling mechanism. This effect would be expected to enhance cardiac contractility. Training lengthened the action potential duration of sub-epicardial myocytes, reducing the transmural gradient in action potential duration. This observation may be important in understanding the cellular causes of T-wave abnormalities found in the electrocardiograms of some athletes. Our study shows that voluntary exercise modulates the morphological, mechanical and electrical properties of cardiac myocytes, and that this modulation is dependent upon the regional origin of the myocytes.
Figures




References
-
- Antzelevitch C, Sicouri S, Litovsky SH, Lukas A, Krishnan SC, Di Diego JM, Gintant GA, Liu D-W. Heterogeneity within the ventricular wall: electrophysiology and pharmacology of epicardial, endocardial and M cells. Circulation Research. 1991;69:1427–1449. - PubMed
-
- Anversa P, Ricci R, Olivetti G. Quantitative structural analysis of the myocardium during physiologic growth and induced cardiac hypertrophy: a review. Journal of the American College of Cardiology. 1986;7:1140–1149. - PubMed
-
- Asami S, Hirano T, Yamaguchi R, Tsurudome Y, Itoh H, Kasai H. Effects of forced and spontaneous exercise on 8-hydroxyguanosine levels in rat organs. Biochemical and Biophysical Research Communications. 1998;24:678–682. - PubMed
-
- Bryant SM, Shipsey J, Hart G. Normal regional distribution of membrane current density in the rat left ventricle is altered in catecholamine-induced hypertrophy. Cardiovascular Research. 1999;42:391–400. - PubMed
-
- Calaghan SC, White E. The role of calcium in the response of cardiac muscle to stretch. Progress in Biophysics and Molecular Biology. 1999;71:1–31. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

