Action spectrum for cryptochrome-dependent hypocotyl growth inhibition in Arabidopsis
- PMID: 12068118
- PMCID: PMC161700
- DOI: 10.1104/pp.010969
Action spectrum for cryptochrome-dependent hypocotyl growth inhibition in Arabidopsis
Abstract
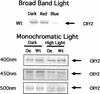
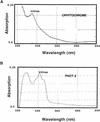
Cryptochrome blue-light photoreceptors are found in both plants and animals and have been implicated in numerous developmental and circadian signaling pathways. Nevertheless, no action spectrum for a physiological response shown to be entirely under the control of cryptochrome has been reported. In this work, an action spectrum was determined in vivo for a cryptochrome-mediated high-irradiance response, the blue-light-dependent inhibition of hypocotyl elongation in Arabidopsis. Comparison of growth of wild-type, cry1cry2 cryptochrome-deficient double mutants, and cryptochrome-overexpressing seedlings demonstrated that responsivity to monochromatic light sources within the range of 390 to 530 nm results from the activity of cryptochrome with no other photoreceptor having a significant primary role at the fluence range tested. In both green- and norflurazon-treated (chlorophyll-deficient) seedlings, cryptochrome activity is fairly uniform throughout its range of maximal response (390-480 nm), with no sharply defined peak at 450 nm; however, activity at longer wavelengths was disproportionately enhanced in CRY1-overexpressing seedlings as compared with wild type. The action spectrum does not correlate well with the absorption spectra either of purified recombinant cryptochrome photoreceptor or to that of a second class of blue-light photoreceptor, phototropin (PHOT1 and PHOT2). Photoreceptor concentration as determined by western-blot analysis showed a greater stability of CRY2 protein under the monochromatic light conditions used in this study as compared with broad band blue light, suggesting a complex mechanism of photoreceptor activation. The possible role of additional photoreceptors (in particular phytochrome A) in cryptochrome responses is discussed.
Figures





References
-
- Ahmad M. Seeing the world in red and blue: insight into plant vision and photoreceptors. Curr Opin Plant Biol. 1999;2:230–235. - PubMed
-
- Ahmad M, Cashmore AR. Seeing blue: the discovery of cryptochrome. Plant Mol Biol. 1996;30:851–861. - PubMed
-
- Ahmad M, Cashmore AR. The blue-light receptor cryptochrome 1 shows functional dependence on phytochrome A or phytochrome B in Arabidopsis thaliana. Plant J. 1997;11:421–427. - PubMed
-
- Ahmad M, Jarillo J, Smirnova O, Cashmore AR. The CRY1 blue light photoreceptor of Arabidopsis interacts with phytochrome A in vitro. Mol Cells. 1998b;1:939–948. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

