Cytokine signaling and hematopoietic homeostasis are disrupted in Lnk-deficient mice
- PMID: 12070287
- PMCID: PMC2193556
- DOI: 10.1084/jem.20011883
Cytokine signaling and hematopoietic homeostasis are disrupted in Lnk-deficient mice
Abstract
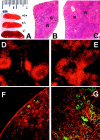
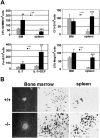
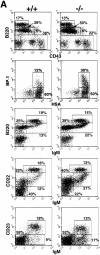
The adaptor protein Lnk, and the closely related proteins APS and SH2B, form a subfamily of SH2 domain-containing proteins implicated in growth factor, cytokine, and immunoreceptor signaling. To elucidate the physiological function of Lnk, we derived Lnk-deficient mice. Lnk(-/-) mice are viable, but display marked changes in the hematopoietic compartment, including splenomegaly and abnormal lymphoid and myeloid homeostasis. The in vitro proliferative capacity and absolute numbers of hematopoietic progenitors from Lnk(-/-) mice are greatly increased, in part due to hypersensitivity to several cytokines. Moreover, an increased synergy between stem cell factor and either interleukin (IL)-3 or IL-7 was observed in Lnk(-/-) cells. Furthermore, Lnk inactivation causes abnormal modulation of IL-3 and stem cell factor-mediated signaling pathways. Consistent with these results, we also show that Lnk is highly expressed in multipotent cells and committed precursors in the erythroid, megakaryocyte, and myeloid lineages. These data implicate Lnk as playing an important role in hematopoiesis and in the regulation of growth factor and cytokine receptor-mediated signaling.
Figures


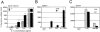





Similar articles
-
Cytokines regulate postnatal hematopoietic stem cell expansion: opposing roles of thrombopoietin and LNK.Genes Dev. 2006 Aug 1;20(15):2018-23. doi: 10.1101/gad.385606. Genes Dev. 2006. PMID: 16882979 Free PMC article.
-
Lnk negatively regulates self-renewal of hematopoietic stem cells by modifying thrombopoietin-mediated signal transduction.Proc Natl Acad Sci U S A. 2007 Feb 13;104(7):2349-54. doi: 10.1073/pnas.0606238104. Epub 2007 Feb 6. Proc Natl Acad Sci U S A. 2007. PMID: 17284614 Free PMC article.
-
Enhanced hematopoiesis by hematopoietic progenitor cells lacking intracellular adaptor protein, Lnk.J Exp Med. 2002 Jan 21;195(2):151-60. doi: 10.1084/jem.20011170. J Exp Med. 2002. PMID: 11805142 Free PMC article.
-
Role of the adaptor protein LNK in normal and malignant hematopoiesis.Oncogene. 2013 Jun 27;32(26):3111-8. doi: 10.1038/onc.2012.435. Epub 2012 Oct 8. Oncogene. 2013. PMID: 23045270 Review.
-
The Lnk adaptor protein: a key regulator of normal and pathological hematopoiesis.Arch Immunol Ther Exp (Warsz). 2012 Dec;60(6):415-29. doi: 10.1007/s00005-012-0194-x. Epub 2012 Sep 19. Arch Immunol Ther Exp (Warsz). 2012. PMID: 22990499 Review.
Cited by
-
Mouse models of diseases of megakaryocyte and platelet homeostasis.Mamm Genome. 2011 Aug;22(7-8):449-65. doi: 10.1007/s00335-011-9336-4. Epub 2011 Jun 11. Mamm Genome. 2011. PMID: 21667128 Review.
-
CBL family E3 ubiquitin ligases control JAK2 ubiquitination and stability in hematopoietic stem cells and myeloid malignancies.Genes Dev. 2017 May 15;31(10):1007-1023. doi: 10.1101/gad.297135.117. Epub 2017 Jun 13. Genes Dev. 2017. PMID: 28611190 Free PMC article.
-
Hematopoietic and eosinophil-specific LNK(SH2B3) deficiency promotes eosinophilia and arterial thrombosis.Blood. 2024 Apr 25;143(17):1758-1772. doi: 10.1182/blood.2023021055. Blood. 2024. PMID: 38096361 Free PMC article.
-
LNK/SH2B3 Loss Exacerbates the Development of Myeloproliferative Neoplasms in CBL-deficient Mice.Stem Cell Rev Rep. 2025 Feb;21(2):509-519. doi: 10.1007/s12015-024-10825-0. Epub 2024 Nov 19. Stem Cell Rev Rep. 2025. PMID: 39560864 Free PMC article.
-
Kinase activation through dimerization by human SH2-B.Mol Cell Biol. 2005 Apr;25(7):2607-21. doi: 10.1128/MCB.25.7.2607-2621.2005. Mol Cell Biol. 2005. PMID: 15767667 Free PMC article.
References
-
- Watowich, S.S., H. Wu, M. Socolovsky, U. Klingmuller, S.N. Constantinescu, and H.F. Lodish. 1996. Cytokine receptor signal transduction and the control of hematopoietic cell development. Annu. Rev. Cell Dev. Biol. 12:91–128. - PubMed
-
- Ihle, J.N. 1995. Cytokine receptor signalling. Nature. 377:591–594. - PubMed
-
- O'Shea, J.J. 1997. Jaks, STATs, cytokine signal transduction, and immunoregulation: are we there yet? Immunity. 7:1–11. - PubMed
-
- Ihle, J.N. 1996. STATs: signal transducers and activators of transcription. Cell. 84:331–334. - PubMed
-
- Ward, A.C., I. Touw, and A. Yoshimura. 2000. The Jak-Stat pathway in normal and perturbed hematopoiesis. Blood. 95:19–29. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

