Time-dependent descending facilitation from the rostral ventromedial medulla maintains, but does not initiate, neuropathic pain
- PMID: 12077208
- PMCID: PMC6757729
- DOI: 10.1523/JNEUROSCI.22-12-05129.2002
Time-dependent descending facilitation from the rostral ventromedial medulla maintains, but does not initiate, neuropathic pain
Abstract
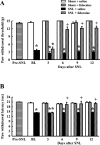
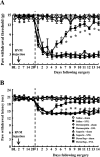
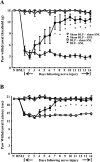
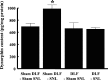
Although injury-induced afferent discharge declines significantly over time, experimental neuropathic pain persists unchanged for long periods. These observations suggest that processes that initiate experimental neuropathic pain may differ from those that maintain such pain. Here, the role of descending facilitation arising from developing plasticity in the rostral ventromedial medulla (RVM) in the initiation and maintenance of experimental neuropathic pain was explored. Tactile and thermal hypersensitivity were induced in rats by spinal nerve ligation (SNL). RVM lidocaine blocked SNL-induced tactile and thermal hypersensitivity on post-SNL days 6-12 but not on post-SNL day 3. Lesion of RVM cells expressing mu-opioid receptors with dermorphin-saporin did not prevent the onset of SNL-induced tactile and thermal hypersensitivity, but these signs reversed to baseline levels beginning on post-SNL day 4. Similarly, lesions of the dorsolateral funiculus (DLF) did not prevent the onset of SNL-induced tactile and thermal hypersensitivity, but these signs reversed to baseline levels beginning on post-SNL day 4. Lesions of the DLF also blocked the SNL-induced increase in spinal dynorphin content, which has been suggested to promote neuropathic pain. These data distinguish mechanisms that initiate the neuropathic state as independent of descending supraspinal influences and additional mechanism(s) that require supraspinal facilitation to maintain such pain. In addition, the data indicate that these time-dependent descending influences can underlie some of the SNL-induced plasticity at the spinal level. Such time-dependent descending influences driving associated spinal changes, such as the upregulation of dynorphin, are key elements in the maintenance, but not initiation, of neuropathic states.
Figures

References
-
- Arcaya JL, Cano G, Gomez G, Maixner W, Suarez-Roca H. Dynorphin A increases substance P release from trigeminal primary afferent C-fibers. Eur J Pharmacol. 1999;366:27–34. - PubMed
-
- Bederson JB, Fields HL, Barbaro NM. Hyperalgesia during naloxone-precipitated withdrawal from morphine is associated with increased on-cell activity in the rostral ventromedial medulla. Somatosens Mot Res. 1990;7:185–203. - PubMed
-
- Bian D, Ossipov MH, Zhong C, Malan TP, Jr, Porreca F. Tactile allodynia, but not thermal hyperalgesia, of the hindlimbs is blocked by spinal transection in rats with nerve injury. Neurosci Lett. 1998;241:79–82. - PubMed
-
- Bian D, Ossipov MH, Ibrahim M, Raffa RB, Tallarida RJ, Malan TP, Jr, Lai J, Porreca F. Loss of antiallodynic and antinociceptive spinal/supraspinal morphine synergy in nerve-injured rats: restoration by MK-801 or dynorphin antiserum. Brain Res. 1999;831:55–63. - PubMed
-
- Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994;53:55–63. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
