Activation of ventral tegmental area cells by the bed nucleus of the stria terminalis: a novel excitatory amino acid input to midbrain dopamine neurons
- PMID: 12077212
- PMCID: PMC6757737
- DOI: 10.1523/JNEUROSCI.22-12-05173.2002
Activation of ventral tegmental area cells by the bed nucleus of the stria terminalis: a novel excitatory amino acid input to midbrain dopamine neurons
Abstract
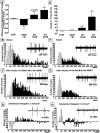
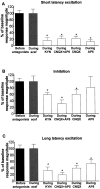
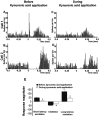
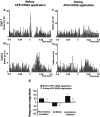
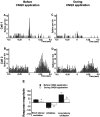
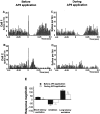
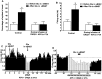
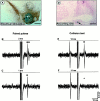
We examined the role of excitatory amino acids (EAAs) in the activation of midbrain dopaminergic (DA) neurons evoked by stimulation of the ventromedial and ventrolateral (subcommissural) bed nucleus of the stria terminalis (vBNST). Using anesthetized rats and extracellular recording techniques, we found that 84.8% of ventral tegmental area (VTA) DA neurons were activated synaptically by single-pulse electrical stimulation of the vBNST. In contrast, similar stimulation did not affect the activity of presumed GABA neurons in the VTA. Three characteristic responses were observed in VTA DA neurons: short latency activation (<25 msec; 55.1% of cells), long latency activation (>65 msec; 56% of cells), and inhibition (61.8% of cells, usually followed by long latency excitation). Microinfusion of antagonists of EAA receptors (3 mm kynurenic acid, 100 microm AP-5, or 50 microm CNQX) from a micropipette adjacent to the recording electrode significantly reduced both short and long latency activations evoked in DA neurons by vBNST stimulation. Specific responses were attenuated similarly by AP-5 alone, CNQX alone, or a cocktail of AP-5+CNQX, indicating that joint activation of NMDA plus non-NMDA receptors was required. Stimulation of the vBNST by local microinfusion of glutamate increased the firing and bursting activity of VTA DA neurons. Similar microinfusion of GABA decreased bursting of VTA DA neurons without altering their firing rate. Retrograde and anterograde labeling and antidromic activation of vBNST neurons by VTA stimulation confirmed a direct projection from the vBNST to the VTA. These results reveal that inputs from the vBNST exert a strong excitatory influence on VTA DA neurons mediated by both NMDA and non-NMDA receptors.
Figures



Similar articles
-
Potent regulation of midbrain dopamine neurons by the bed nucleus of the stria terminalis.J Neurosci. 2001 Aug 15;21(16):RC160. doi: 10.1523/JNEUROSCI.21-16-j0003.2001. J Neurosci. 2001. PMID: 11473131 Free PMC article.
-
Regulation of the ventral tegmental area by the bed nucleus of the stria terminalis is required for expression of cocaine preference.Eur J Neurosci. 2012 Dec;36(11):3549-58. doi: 10.1111/j.1460-9568.2012.08277.x. Epub 2012 Oct 8. Eur J Neurosci. 2012. PMID: 23039920 Free PMC article.
-
Role of excitatory amino acids in the ventral tegmental area for central actions of non-competitive NMDA-receptor antagonists and nicotine.Amino Acids. 1998;14(1-3):51-6. doi: 10.1007/BF01345242. Amino Acids. 1998. PMID: 9871441
-
Anatomy and Function of Ventral Tegmental Area Glutamate Neurons.Front Neural Circuits. 2022 May 20;16:867053. doi: 10.3389/fncir.2022.867053. eCollection 2022. Front Neural Circuits. 2022. PMID: 35669454 Free PMC article. Review.
-
A thalamo-hippocampal-ventral tegmental area loop may produce the positive feedback that underlies the psychotic break in schizophrenia.Biol Psychiatry. 2010 Jul 1;68(1):17-24. doi: 10.1016/j.biopsych.2010.04.007. Epub 2010 May 31. Biol Psychiatry. 2010. PMID: 20553749 Free PMC article. Review.
Cited by
-
Genetic dissection of midbrain dopamine neuron development in vivo.Dev Biol. 2012 Dec 15;372(2):249-62. doi: 10.1016/j.ydbio.2012.09.019. Epub 2012 Oct 4. Dev Biol. 2012. PMID: 23041116 Free PMC article.
-
Acute and chronic methylphenidate administration in intact and VTA-specific and nonspecific lesioned rats.J Neural Transm (Vienna). 2019 Feb;126(2):173-182. doi: 10.1007/s00702-018-1963-4. Epub 2019 Jan 8. J Neural Transm (Vienna). 2019. PMID: 30617502
-
Chemogenetic Manipulations of Ventral Tegmental Area Dopamine Neurons Reveal Multifaceted Roles in Cocaine Abuse.J Neurosci. 2019 Jan 16;39(3):503-518. doi: 10.1523/JNEUROSCI.0537-18.2018. Epub 2018 Nov 16. J Neurosci. 2019. PMID: 30446532 Free PMC article.
-
Properties of distinct ventral tegmental area synapses activated via pedunculopontine or ventral tegmental area stimulation in vitro.J Physiol. 2009 Mar 15;587(Pt 6):1233-47. doi: 10.1113/jphysiol.2008.164194. Epub 2009 Feb 2. J Physiol. 2009. PMID: 19188251 Free PMC article.
-
α(2A)-adrenergic receptors filter parabrachial inputs to the bed nucleus of the stria terminalis.J Neurosci. 2014 Jul 9;34(28):9319-31. doi: 10.1523/JNEUROSCI.0822-14.2014. J Neurosci. 2014. PMID: 25009265 Free PMC article.
References
-
- Acquas E, Di Chiara G. Depression of mesolimbic dopamine transmission and sensitization to morphine during opiate abstinence. J Neurochem. 1992;58:1620–1625. - PubMed
-
- Aghajanian GK, Bunney BS. Dopamine “autoreceptors”: pharmacological characterization by microiontophoretic single cell recording studies. Naunyn Schmiedebergs Arch Pharmacol. 1977;297:1–7. - PubMed
-
- Aston-Jones G, Shaver R, Dinan TG. Nucleus basalis neurons exhibit axonal branching with decreased impulse conduction velocity in rat cerebrocortex. Brain Res. 1985;325:271–285. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
