Neither agouti-related protein nor neuropeptide Y is critically required for the regulation of energy homeostasis in mice
- PMID: 12077332
- PMCID: PMC139785
- DOI: 10.1128/MCB.22.14.5027-5035.2002
Neither agouti-related protein nor neuropeptide Y is critically required for the regulation of energy homeostasis in mice
Abstract
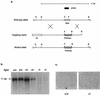
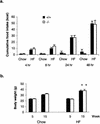
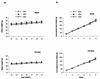
Agouti-related protein (AgRP), a neuropeptide abundantly expressed in the arcuate nucleus of the hypothalamus, potently stimulates feeding and body weight gain in rodents. AgRP is believed to exert its effects through the blockade of signaling by alpha-melanocyte-stimulating hormone at central nervous system (CNS) melanocortin-3 receptor (Mc3r) and Mc4r. We generated AgRP-deficient (Agrp(-/-)) mice to examine the physiological role of AgRP. Agrp(-/-) mice are viable and exhibit normal locomotor activity, growth rates, body composition, and food intake. Additionally, Agrp(-/-) mice display normal responses to starvation, diet-induced obesity, and the administration of exogenous leptin or neuropeptide Y (NPY). In situ hybridization failed to detect altered CNS expression levels for proopiomelanocortin, Mc3r, Mc4r, or NPY mRNAs in Agrp(-/-) mice. As AgRP and the orexigenic peptide NPY are coexpressed in neurons of the arcuate nucleus, we generated AgRP and NPY double-knockout (Agrp(-/-);Npy(-/-)) mice to determine whether NPY or AgRP plays a compensatory role in Agrp(-/-) or NPY-deficient (Npy(-/-)) mice, respectively. Similarly to mice deficient in either AgRP or NPY, Agrp(-/-);Npy(-/-) mice suffer no obvious feeding or body weight deficits and maintain a normal response to starvation. Our results demonstrate that neither AgRP nor NPY is a critically required orexigenic factor, suggesting that other pathways capable of regulating energy homeostasis can compensate for the loss of both AgRP and NPY.
Figures





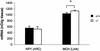

References
-
- Bultman, S. J., E. J. Michaud, and R. P. Woychik. 1992. Molecular characterization of the mouse agouti locus. Cell 71:1195-1204. - PubMed
-
- Butler, A. A., R. A. Kesterson, K. Khong, M. J. Cullen, M. A. Pelleymounter, J. Dekoning, M. Baetscher, and R. D. Cone. 2000. A unique metabolic syndrome causes obesity in the melanocortin-3 receptor-deficient mouse. Endocrinology 141:3518-3521. - PubMed
-
- Chen, A. S., D. J. Marsh, M. E. Trumbauer, E. G. Frazier, X. M. Guan, H. Yu, C. I. Rosenblum, A. Vongs, Y. Feng, L. Cao, J. M. Metzger, A. M. Strack, R. E. Camacho, T. N. Mellin, C. N. Nunes, W. Min, J. Fisher, S. Gopal-Truter, D. E. MacIntyre, H. Y. Chen, and L. H. T. Van der Ploeg. 2000. Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nat. Genet. 26:97-102. - PubMed
-
- Chen, A. S., J. M. Metzger, M. E. Trumbauer, X. M. Guan, H. Yu, E. G. Frazier, D. J. Marsh, M. J. Forrest, S. Gopal-Truter, J. Fisher, R. E. Camacho, A. M. Strack, T. N. Mellin, D. E. MacIntyre, H. Y. Chen, and L. H. T. Van der Ploeg. 2000. Role of melanocortin-4 receptor in metabolic rate and food intake in mice. Transgenic Res. 9:145-154. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
