Transmission of cell stress from endoplasmic reticulum to mitochondria: enhanced expression of Lon protease
- PMID: 12082077
- PMCID: PMC2173558
- DOI: 10.1083/jcb.200108103
Transmission of cell stress from endoplasmic reticulum to mitochondria: enhanced expression of Lon protease
Abstract
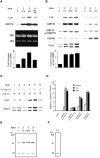
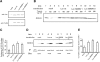
The rat homologue of a mitochondrial ATP-dependent protease Lon was cloned from cultured astrocytes exposed to hypoxia. Expression of Lon was enhanced in vitro by hypoxia or ER stress, and in vivo by brain ischemia. These observations suggested that changes in nuclear gene expression (Lon) triggered by ER stress had the potential to impact important mitochondrial processes such as assembly and/or degradation of cytochrome c oxidase (COX). In fact, steady-state levels of nuclear-encoded COX IV and V were reduced, and mitochondrial-encoded subunit II was rapidly degraded under ER stress. Treatment of cells with cycloheximide caused a similar imbalance in the accumulation of COX subunits, and enhanced mRNA for Lon and Yme1, the latter another mitochondrial ATP-dependent protease. Furthermore, induction of Lon or GRP75/mtHSP70 by ER stress was inhibited in PERK (-/-) cells. Transfection studies revealed that overexpression of wild-type or proteolytically inactive Lon promoted assembly of COX II into a COX I-containing complex, and partially prevented mitochondrial dysfunction caused by brefeldin A or hypoxia. These observations demonstrated that suppression of protein synthesis due to ER stress has a complex effect on the synthesis of mitochondrial-associated proteins, both COX subunits and ATP-dependent proteases and/or chaperones contributing to assembly of the COX complex.
Figures





Similar articles
-
Promotion of mitochondrial membrane complex assembly by a proteolytically inactive yeast Lon.Science. 1996 Oct 4;274(5284):103-6. doi: 10.1126/science.274.5284.103. Science. 1996. PMID: 8810243
-
Mitochondrial LON protease-dependent degradation of cytochrome c oxidase subunits under hypoxia and myocardial ischemia.Biochim Biophys Acta Bioenerg. 2017 Jul;1858(7):519-528. doi: 10.1016/j.bbabio.2017.04.003. Epub 2017 Apr 23. Biochim Biophys Acta Bioenerg. 2017. PMID: 28442264 Free PMC article.
-
The ATPase and protease domains of yeast mitochondrial Lon: roles in proteolysis and respiration-dependent growth.Proc Natl Acad Sci U S A. 1998 Sep 1;95(18):10584-9. doi: 10.1073/pnas.95.18.10584. Proc Natl Acad Sci U S A. 1998. PMID: 9724747 Free PMC article.
-
Upregulation of the mitochondrial Lon Protease allows adaptation to acute oxidative stress but dysregulation is associated with chronic stress, disease, and aging.Redox Biol. 2013 Feb 9;1(1):258-64. doi: 10.1016/j.redox.2013.01.015. Redox Biol. 2013. PMID: 24024159 Free PMC article. Review.
-
Lon in maintaining mitochondrial and endoplasmic reticulum homeostasis.Arch Toxicol. 2018 Jun;92(6):1913-1923. doi: 10.1007/s00204-018-2210-3. Epub 2018 May 2. Arch Toxicol. 2018. PMID: 29721585 Review.
Cited by
-
Inhibition of Lon blocks cell proliferation, enhances chemosensitivity by promoting apoptosis and decreases cellular bioenergetics of bladder cancer: potential roles of Lon as a prognostic marker and therapeutic target in baldder cancer.Oncotarget. 2014 Nov 30;5(22):11209-24. doi: 10.18632/oncotarget.2026. Oncotarget. 2014. PMID: 25526030 Free PMC article.
-
Phosphorylation of human TFAM in mitochondria impairs DNA binding and promotes degradation by the AAA+ Lon protease.Mol Cell. 2013 Jan 10;49(1):121-32. doi: 10.1016/j.molcel.2012.10.023. Epub 2012 Nov 29. Mol Cell. 2013. PMID: 23201127 Free PMC article.
-
Impact of Carcinogenic Chromium on the Cellular Response to Proteotoxic Stress.Int J Mol Sci. 2019 Oct 3;20(19):4901. doi: 10.3390/ijms20194901. Int J Mol Sci. 2019. PMID: 31623305 Free PMC article. Review.
-
Targeting Cancer with Phytochemicals via Their Fine Tuning of the Cell Survival Signaling Pathways.Int J Mol Sci. 2018 Nov 12;19(11):3568. doi: 10.3390/ijms19113568. Int J Mol Sci. 2018. PMID: 30424557 Free PMC article. Review.
-
Intercepting the Lipid-Induced Integrated Stress Response Reduces Atherosclerosis.J Am Coll Cardiol. 2019 Mar 19;73(10):1149-1169. doi: 10.1016/j.jacc.2018.12.055. J Am Coll Cardiol. 2019. PMID: 30871699 Free PMC article.
References
-
- Amerik, A.Y., G.V. Petukhova, V.G. Grigorenko, I.P. Lykov, S.V. Yarovoi, V.M. Lipkin, and A.E. Gorbalenya. 1994. Cloning and sequence analysis of cDNA for a human homologue of eubacterial ATP-dependent Lon proteases. FEBS Lett. 340:25–28. - PubMed
-
- Casari, G., M. De Fusco, S. Ciarmatori, M. Zeviani, M. Mora, P. Fernandez, G. De Michele, A. Filla, S. Cocozza, R. Marconi, A. Dürr, B. Fontaine, and A. Ballabio. 1998. Spastic paraplegia and OXPHOS impairment caused by mutation in paraplegin, a nuclear-encoded mitochondrial metalloprotease. Cell. 93:973–983. - PubMed
-
- Evans, W.H., 1992. Preparative Centrifugation. A Practical Approach. Rickwood, D., editor. Oxford University Press, Oxford, UK. 399 pp.
-
- Kobara, M., T. Tatsumi, S. Matoba, Y. Yamahara, C. Nakagawa, B. Ohta, T. Matumoto, D. Inoue, J. Asayama, and M. Nakagawa. 1996. Effect of ischemic preconditioning on mitochondrial oxidative phosphorylation and high energy phosphates in rat hearts. J. Mol. Cell. Cardiol. 28:417–428. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

