Microarray and biochemical analysis of lovastatin-induced apoptosis of squamous cell carcinomas
- PMID: 12082550
- PMCID: PMC1531704
- DOI: 10.1038/sj.neo.7900247
Microarray and biochemical analysis of lovastatin-induced apoptosis of squamous cell carcinomas
Abstract
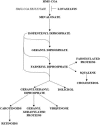
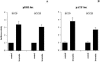
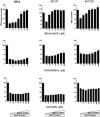
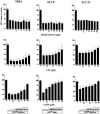
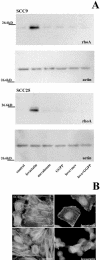
We recently identified 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, the rate-limiting enzyme of the mevalonate pathway, as a potential therapeutic target of the head and neck squamous cell carcinomas (HNSCC) and cervical carcinomas (CC). The products of this complex biochemical pathway, including de novo cholesterol, are vital for a variety of key cellular functions affecting membrane integrity, cell signaling, protein synthesis, and cell cycle progression. Lovastatin, a specific inhibitor of HMG-CoA reductase, induces a pronounced apoptotic response in a specific subset of tumor types, including HNSCC and CC. The mediators of this response are not well established. Identification of differentially expressed genes represents a feasible approach to delineate these mediators as lovastatin has the potential to modulate transcription indirectly by perturbing levels of sterols and other mevalonate metabolites. Expression analysis following treatment of the HNSCC cell lines SCC9 or SCC25 with 10 microM lovastatin for 1 day showed that less than 2% (9 cDNAs) of the 588 cDNAs on this microarray were affected in both cell lines. These included diazepam-binding inhibitor/acyl-CoA-binding protein, the activated transcription factor 4 and rhoA. Because the biosynthesis of mevalonate leads to its incorporation into more than a dozen classes of end products, their role in lovastatin-induced apoptosis was also evaluated. Addition of the metabolites of all the major branches of the mevalonate pathway indicated that only the nonsterol moiety, geranylgeranyl pyrophosphate (GGPP), significantly inhibited the apoptotic effects of lovastatin in HNSCC and CC cells. Because rhoA requires GGPP for its function, this links the microarray and biochemical data and identifies rhoA as a potential mediator of the anticancer properties of lovastatin. Our data suggest that the depletion of nonsterol mevalonate metabolites, particularly GGPP, can be potential mediators of lovastatin-induced apoptosis of HNSCC and CC cells.
Figures
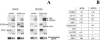
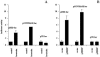
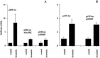
References
-
- Agarwal B, Rao CV, Bhendwal S, Ramey WR, Shirin H, Reddy BS, Holt PR. Lovastatin augments sulindac-induced apoptosis in colon cancer cells and potentiates chemopreventive effect of sulindac. Proc Am Assoc Cancer Res. 1999;40:57. - PubMed
-
- Aznar S, Lacal JC. Rho signals to cell growth and apoptosis. Cancer Lett. 2001;165:1–10. - PubMed
-
- Corsini A, Maggi FM, Catapano AL. Pharmacology of competitive inhibitors of HMG-CoA reductase. Pharmacol Res. 1995;31:9–27. - PubMed
-
- Cunningham MJ. Genomics and proteomics: the new millennium of drug discovery and development. J Pharmacol Toxicol Methods. 2000;44:291–300. - PubMed
