Influence of the electric charge of the antigen and the immune complex (IC) lattice on the IC activation of human complement
- PMID: 12084047
- PMCID: PMC2517671
- DOI: 10.1046/j.1365-2613.2002.00224.x
Influence of the electric charge of the antigen and the immune complex (IC) lattice on the IC activation of human complement
Abstract
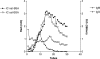
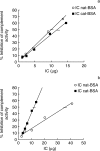
In order to understand the mechanism of complement (C) activation by immune complexes (ICs), the anti-complementary effect of ICs containing cationized antigens was compared in vitro to that using ICs formed by native antigens. ICs were prepared with affinity-purified rabbit polyclonal IgG antibovine serum albumin (BSA) antibody and either native BSA (isoelectric point 4.2) or BSA rendered cationic by treatment with ethylenediamine (isoelectric point 9.4). Native and cationized antigens were characterized by isoelectric focusing. ICs containing anti-BSA IgG or F(ab')2, formed either at equivalence or in excess of native or cationized antigen, were submitted to ultracentrifugation in a sucrose gradient for mesh size determination. The anti-complementary effect of ICs was evaluated by kinetic determination of haemolytic activity of human serum on haemolysin-sensitized sheep red blood cells. In conditions of antigen excess, the ICs formed by cationized BSA were significantly more efficient in activating human complement than those formed by native antigen. This higher activity was dependent on cationized antigen complexed with complete antibody molecules, as non-complexed cationized BSA or ICs prepared with F(ab')2 fragments were inactive under the same experimental conditions. Furthermore, this difference did not depend on the mesh size of the immune complexes. Our results suggest that the balance between antigen, antibody and C may be of importance in vivo for the onset and course of infections and other pathological processes involving IC formation. ICs containing cationized antigens should be proven of value in experimental models for studies on the regulation of C activation.
Figures

References
-
- Barbosa JE. Determinação cinética de atividade anticomplementar PhD Thesis. Faculdade de Medicina de Ribeirão Preto. Universidade de São Paulo; 1983.
-
- Buyon JP, Zuckermann JD. Articular manifestation of systemic lupus erythematosus. In: Lahita RG, editor. Systemic Lupus Erythematosus. London: Churchill Livingstone; 1987. pp. 791–799.
-
- Caughman GB, Boackle RJ, Vesely J. A postulated mechanism for heparin's potentiation of C1 inhibitor function. Mol. Immunol. 1982;19:287–295. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

