Spontaneous transient outward currents arise from microdomains where BK channels are exposed to a mean Ca(2+) concentration on the order of 10 microM during a Ca(2+) spark
- PMID: 12084772
- PMCID: PMC2311396
- DOI: 10.1085/jgp.20028571
Spontaneous transient outward currents arise from microdomains where BK channels are exposed to a mean Ca(2+) concentration on the order of 10 microM during a Ca(2+) spark
Abstract
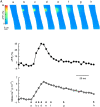
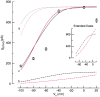
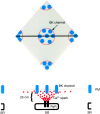
Ca(2+) sparks are small, localized cytosolic Ca(2+) transients due to Ca(2+) release from sarcoplasmic reticulum through ryanodine receptors. In smooth muscle, Ca(2+) sparks activate large conductance Ca(2+)-activated K(+) channels (BK channels) in the spark microdomain, thus generating spontaneous transient outward currents (STOCs). The purpose of the present study is to determine experimentally the level of Ca(2+) to which the BK channels are exposed during a spark. Using tight seal, whole-cell recording, we have analyzed the voltage-dependence of the STOC conductance (g((STOC))), and compared it to the voltage-dependence of BK channel activation in excised patches in the presence of different [Ca(2+)]s. The Ca(2+) sparks did not change in amplitude over the range of potentials of interest. In contrast, the magnitude of g((STOC)) remained roughly constant from 20 to -40 mV and then declined steeply at more negative potentials. From this and the voltage dependence of BK channel activation, we conclude that the BK channels underlying STOCs are exposed to a mean [Ca(2+)] on the order of 10 microM during a Ca(2+) spark. The membrane area over which a concentration > or =10 microM is reached has an estimated radius of 150-300 nm, corresponding to an area which is a fraction of one square micron. Moreover, given the constraints imposed by the estimated channel density and the Ca(2+) current during a spark, the BK channels do not appear to be uniformly distributed over the membrane but instead are found at higher density at the spark site.
Figures





Similar articles
-
Dynamics of signaling between Ca(2+) sparks and Ca(2+)- activated K(+) channels studied with a novel image-based method for direct intracellular measurement of ryanodine receptor Ca(2+) current.J Gen Physiol. 2000 Dec;116(6):845-64. doi: 10.1085/jgp.116.6.845. J Gen Physiol. 2000. PMID: 11099351 Free PMC article.
-
Local Ca(2+) transients and distribution of BK channels and ryanodine receptors in smooth muscle cells of guinea-pig vas deferens and urinary bladder.J Physiol. 2001 Jul 15;534(Pt. 2):313-26. doi: 10.1111/j.1469-7793.2001.t01-3-00313.x. J Physiol. 2001. PMID: 11454953 Free PMC article.
-
Ca(2+) spark sites in smooth muscle cells are numerous and differ in number of ryanodine receptors, large-conductance K(+) channels, and coupling ratio between them.Am J Physiol Cell Physiol. 2004 Dec;287(6):C1577-88. doi: 10.1152/ajpcell.00153.2004. Epub 2004 Aug 11. Am J Physiol Cell Physiol. 2004. PMID: 15306542
-
Ca2+ signalling and Ca2+-activated K+ channels in smooth muscle.Novartis Found Symp. 2002;246:52-64; discussion 64-70, 221-7. Novartis Found Symp. 2002. PMID: 12164315 Review.
-
Signaling between SR and plasmalemma in smooth muscle: sparks and the activation of Ca2+-sensitive ion channels.Cell Calcium. 2003 Sep;34(3):211-29. doi: 10.1016/s0143-4160(03)00124-6. Cell Calcium. 2003. PMID: 12887969 Review.
Cited by
-
Spatial organization of RYRs and BK channels underlying the activation of STOCs by Ca(2+) sparks in airway myocytes.J Gen Physiol. 2011 Aug;138(2):195-209. doi: 10.1085/jgp.201110626. Epub 2011 Jul 11. J Gen Physiol. 2011. PMID: 21746845 Free PMC article.
-
Ca2+-sparks constitute elementary building blocks for global Ca2+-signals in myocytes of retinal arterioles.Cell Calcium. 2007 May;41(5):451-66. doi: 10.1016/j.ceca.2006.08.005. Epub 2006 Oct 5. Cell Calcium. 2007. PMID: 17027081 Free PMC article.
-
Syntillas release Ca2+ at a site different from the microdomain where exocytosis occurs in mouse chromaffin cells.Biophys J. 2006 Mar 15;90(6):2027-37. doi: 10.1529/biophysj.105.071654. Epub 2005 Dec 30. Biophys J. 2006. PMID: 16387759 Free PMC article.
-
Differential contribution of Ca2+ sources to day and night BK current activation in the circadian clock.J Gen Physiol. 2018 Feb 5;150(2):259-275. doi: 10.1085/jgp.201711945. Epub 2017 Dec 13. J Gen Physiol. 2018. PMID: 29237755 Free PMC article.
-
Ca2+-Activated K+ Channels and the Regulation of the Uteroplacental Circulation.Int J Mol Sci. 2023 Jan 10;24(2):1349. doi: 10.3390/ijms24021349. Int J Mol Sci. 2023. PMID: 36674858 Free PMC article. Review.
References
-
- Arnaudeau, S., F.X. Boittin, N. Macrez, J.L. Lavie, C. Mironneau, and J. Mironneau. 1997. L-type and Ca2+ release channel-dependent hierarchical Ca2+ signaling in rat portal vein myocytes. Cell Calcium. 22:399–411. - PubMed
-
- Becker, P.L., J.J. Singer, J.V. Walsh, Jr., and F.S. Fay. 1989. Regulation of calcium concentration in voltage-clamped single smooth muscle cells. Science. 244:211–214. - PubMed
-
- Bezprozvanny, I., J. Watras, and B.E. Ehrlich. 1991. Bell-shaped calcium-response curves of Ins(1,4,5)P3- and calcium-gated channels from endoplasmic reticulum of cerebellum. Nature. 351:751–754. - PubMed
-
- Bolton, T.B., and D.V. Gordienko. 1998. Confocal imaging of calcium release events in single smooth muscle cells. Acta Physiol. Scand. 164:567–575. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

