Dark-stimulated calcium ion fluxes in the chloroplast stroma and cytosol
- PMID: 12084827
- PMCID: PMC150780
- DOI: 10.1105/tpc.000653
Dark-stimulated calcium ion fluxes in the chloroplast stroma and cytosol
Abstract
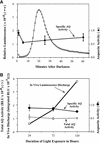
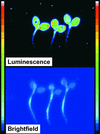
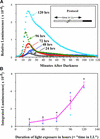
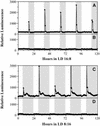
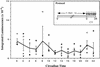
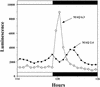
Using transgenic Nicotiana plumbaginifolia seedlings in which the calcium reporter aequorin is targeted to the chloroplast stroma, we found that darkness stimulates a considerable flux of Ca(2+) into the stroma. This Ca(2+) flux did not occur immediately after the light-to-dark transition but began approximately 5 min after lights off and increased to a peak at approximately 20 to 30 min after the onset of darkness. Imaging of aequorin emission confirmed that the dark-stimulated luminescence emanated from chloroplast-containing tissues of the seedling. The magnitude of the Ca(2+) flux was proportional to the duration of light exposure (24 to 120 h) before lights off; the longer the duration of light exposure, the larger the dark-stimulated Ca(2+) flux. On the other hand, the magnitude of the dark-stimulated Ca(2+) flux did not appear to vary as a function of circadian time. When seedlings were maintained on a 24-h light/dark cycle, there was a stromal Ca(2+) burst after lights off every day. Moreover, the waveform of the Ca(2+) spike was different during long-day versus short-day light/dark cycles. The dark-stimulated Ca(2+) flux into the chloroplastidic stroma appeared to affect transient changes in cytosolic Ca(2+) levels. DCMU, an inhibitor of photosynthetic electron transport, caused a significant increase in stromal Ca(2+) levels in the light but did not affect the magnitude of the dark-stimulated Ca(2+) flux. This robust Ca(2+) flux likely plays regulatory roles in the sensing of both light/dark transitions and photoperiod.
Figures


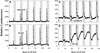
References
-
- Becker, D.W., Callahan, F.E., and Cheniae, G.M. (1985). Photoactivation of NH2OH-treated leaves: Reassembly of released extrinsic PSII polypeptides and religation of Mn into the polynuclear Mn catalyst of water oxidation. FEBS Lett. 192, 209–214.
-
- Bush, D.S. (1995). Calcium regulation in plant cells and its role in signaling. Annu. Rev. Plant Physiol. Plant Mol. Biol. 46, 95–122.
-
- Demmig, B., and Gimmler, H. (1979). Effect of divalent cations on cation fluxes across the chloroplast envelope and on photosynthesis of intact chloroplasts. Z. Naturforsch. 34c, 233–241.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

