Implication of mitochondria-derived ROS and cardiolipin peroxidation in N-(4-hydroxyphenyl)retinamide-induced apoptosis
- PMID: 12085192
- PMCID: PMC2375439
- DOI: 10.1038/sj.bjc.6600356
Implication of mitochondria-derived ROS and cardiolipin peroxidation in N-(4-hydroxyphenyl)retinamide-induced apoptosis
Abstract
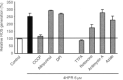
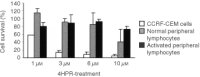
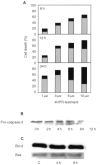
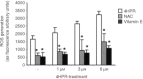
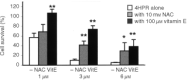
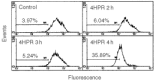
We have studied the effect of N-(4-hydroxyphenyl)retinamide on either malignant human leukaemia cells or normal cells and investigated its mechanism of action. We demonstrate that 4HPR induces reactive oxygen species increase on mitochondria at a target between mitochondrial respiratory chain complex I and II. Such oxidative stress causes cardiolipin peroxidation which in turn allows cytochrome c release to cytosol, caspase-3 activation and therefore apoptotic consumption. Moreover, this apoptotic pathway seems to be bcl-2/bax independent and count only on malignant cells but not normal nor activated lymphocytes.
Copyright 2002 Cancer Research UK
Figures


References
-
- DeliaDAielloALombardiLPelicciGGrignaniFGrignaniFFormelliFMenardSCostaSCostaAVeronesiUPierottiMA1993N-(4-Hydroxyphenyl)retinamide induces apoptosis of malignant hemopoietic cell lines including those unresponsive to retinoic acid Cancer Res 5360366041 - PubMed
-
- DeliaDAielloAMeroniLNicoliniMReedJCPierottiMA1997Role of antioxidants and intracellular free radicals in retinamide-induced cell death Carcinogenesis 18943948 - PubMed
-
- Fernández-ChecaJCGarcía-RuizCColellAMoralesAMaríMMirandaMArditeE1998Oxidative stress: role of mitochondria and protection by glutathione BioFactors 8711 - PubMed
-
- FormelliFClericiMCampaTDi MauroMGMagniAMascottiG1993Five-year administration of fenretinide: pharmacokinetics and effects on plasma retinol concentrations J Clin Oncol 1120362042 - PubMed
-
- HailNLotanR2001Mitochondrial respiration is uniquely associated with the prooxidant and apoptotic effects of N-(4-hydroxyphenyl)retinamide J Biol Chem 2764561445621 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

