Induction of ovarian cancer by defined multiple genetic changes in a mouse model system
- PMID: 12086888
- PMCID: PMC2267863
- DOI: 10.1016/s1535-6108(01)00002-2
Induction of ovarian cancer by defined multiple genetic changes in a mouse model system
Abstract
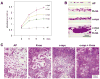
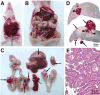
We have developed a mouse model for ovarian carcinoma by using an avian retroviral gene delivery technique for the introduction of multiple genes into somatic ovarian cells of adult mice. Ovarian cells from transgenic mice engineered to express the gene encoding the avian receptor TVA were efficiently infected in vitro with multiple vectors carrying coding sequences for oncogenes and marker genes. When target cells were derived from TVA transgenic mice deficient for p53, the addition of any two of the oncogenes c-myc, K-ras, and Akt were sufficient to induce ovarian tumor formation when infected cells were injected at subcutaneous, intraperitoneal, or ovarian sites. We demonstrated that the ovarian surface epithelium is the precursor tissue for these ovarian carcinomas, and that introduction of oncogenes causes phenotypic changes in the ovarian surface epithelial cells. The induced ovarian tumors in mice resembled human ovarian carcinomas in their rapid progression and intraperitoneal metastatic spread.
Figures




Comment in
-
Of mice and women.Cancer Cell. 2002 Feb;1(1):11-2. doi: 10.1016/s1535-6108(02)00021-1. Cancer Cell. 2002. PMID: 12086881
References
-
- Adams AT, Auersperg N. Transformation of cultured rat ovarian surface epithelial cells by Kirsten murine sarcoma virus. Cancer Res. 1981;41:2063–2072. - PubMed
-
- Auersperg N, Wong AS, Choi KC, Kang SK, Leung PC. Ovarian surface epithelium: biology, endocrinology, and pathology. Endocr Rev. 2001;22:255–288. - PubMed
-
- Aunoble B, Sanches R, Didier E, Bignon YJ. Major oncogenes and tumor suppressor genes involved in epithelial ovarian cancer (review) Int J Oncol. 2000;16:567–576. - PubMed
-
- Baker VV, Borst MP, Dixon D, Hatch KD, Shingleton HM, Miller D. c-myc amplification in ovarian cancer. Gynecol Oncol. 1990;38:340–342. - PubMed
-
- Bates P, Young JA, Varmus HE. A receptor for subgroup A Rous sarcoma virus is related to the low density lipoprotein receptor. Cell. 1993;74:1043–1051. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

