Early kinetic intermediate in the folding of acyl-CoA binding protein detected by fluorescence labeling and ultrarapid mixing
- PMID: 12096190
- PMCID: PMC125024
- DOI: 10.1073/pnas.152321499
Early kinetic intermediate in the folding of acyl-CoA binding protein detected by fluorescence labeling and ultrarapid mixing
Abstract
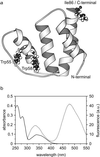
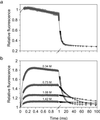
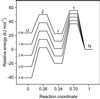
Early conformational events during folding of acyl-CoA binding protein (ACBP), an 86-residue alpha-helical protein, were explored by using a continuous-flow mixing apparatus with a dead time of 70 micros to measure changes in intrinsic tryptophan fluorescence and tryptophan-dansyl fluorescence energy transfer. Although the folding of ACBP was initially described as a concerted two-state process, the tryptophan fluorescence measurements revealed a previously unresolved phase with a time constant tau = 80 micros, indicating formation of an intermediate with only slightly enhanced fluorescence of Trp-55 and Trp-58 relative to the unfolded state. To amplify this phase, a dansyl fluorophore was introduced at the C terminus by labeling an I86C mutant of ACBP with 5-IAEDANS [5-((((2-iodoacetyl)amino)ethyl)amino)naphthalene-1-sulfonic acid]. Continuous-flow refolding of guanidine HCl-denatured ACBP showed a major increase in tryptophan-dansyl fluorescence energy transfer, indicating formation of a partially collapsed ensemble of states on the 100-micros time scale. A subsequent decrease in dansyl fluorescence is attributed to intramolecular quenching of donor fluorescence on formation of the native state. The kinetic data are fully accounted for by three-state mechanisms with either on- or off-pathway intermediates. The intermediate accumulates to a maximum population of 40%, and its stability depends only weakly on denaturant concentration, which is consistent with a marginally stable ensemble of partially collapsed states with approximately 1/3 of the solvent-accessible surface buried. The findings indicate that ultrafast mixing methods combined with sensitive conformational probes can reveal transient accumulation of intermediate states in proteins with apparent two-state folding mechanisms.
Figures

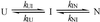

References
-
- Park S-H, Shastry M C R, Roder H. Nat Struct Biol. 1999;6:943–947. - PubMed
-
- Dalby P A, Oliveberg M, Fersht A R. J Mol Biol. 1998;276:625–646. - PubMed
-
- Raschke T M, Marqusee S. Nat Struct Biol. 1997;4:298–304. - PubMed
-
- Capaldi A P, Shastry M C, Kleanthous C, Roder H, Radford S E. Nat Struct Biol. 2001;8:68–72. - PubMed
-
- Jackson S E, Fersht A R. Biochemistry. 1991;30:10428–10435. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

