Differential graft-versus-leukaemia effect by CD28 and CD40 co-stimulatory blockade after graft-versus-host disease prophylaxis
- PMID: 12100023
- PMCID: PMC1906426
- DOI: 10.1046/j.1365-2249.2002.01857.x
Differential graft-versus-leukaemia effect by CD28 and CD40 co-stimulatory blockade after graft-versus-host disease prophylaxis
Abstract
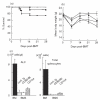
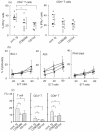
Co-stimulatory blockade may be a promising strategy for tolerance induction in transplantation. In allogeneic bone marrow transplantation (BMT) for leukaemia treatment, however, preservation of the graft-versus-leukaemia (GVL) effect is another critical requirement for clinical application. In this study, we have compared the effect on GVL of using CD28 and CD40 co-stimulatory blockades as graft-versus-host disease (GVHD) prophylaxis in a murine allogeneic BMT model with simultaneous transfer of BCL1 leukaemia. Despite the relative improvement of GVHD as assessed by survival and body weight in both treatment regimes, treatment with anti-CD154 moAb clearly diminished the GVL effect, whereas treatment with anti-CD80 and CD86 MoAbs maintained this effect. Although T cell-mediated effector function at 14 days post-BMT assessed by IFNgamma expression and cytotoxicity against host alloantigen was comparable between both co-stimulatory blockades, IL-12 mRNA expression was preferentially reduced by CD40 blockade. Our results suggest the differential involvement of the CD28 and CD40 co-stimulatory pathways in the development of GVHD and GVL effects. CD28 blockade may be a favourable strategy for tolerance induction in leukaemia patients undergoing BMT.
Figures


Similar articles
-
CD40 ligand-specific antibodies synergize with cyclophosphamide to promote long-term transplantation tolerance across MHC barriers but inhibit graft-vs-leukemia effects of transplanted cells.Exp Hematol. 2003 Jan;31(1):81-8. doi: 10.1016/s0301-472x(02)01009-3. Exp Hematol. 2003. PMID: 12543110
-
Roles of CD28, CTLA4, and inducible costimulator in acute graft-versus-host disease in mice.Biol Blood Marrow Transplant. 2011 Jul;17(7):962-9. doi: 10.1016/j.bbmt.2011.01.018. Epub 2011 Mar 27. Biol Blood Marrow Transplant. 2011. PMID: 21447398 Free PMC article.
-
Host CD40 ligand deficiency induces long-term allograft survival and donor-specific tolerance in mouse cardiac transplantation but does not prevent graft arteriosclerosis.J Immunol. 2000 Sep 15;165(6):3506-18. doi: 10.4049/jimmunol.165.6.3506. J Immunol. 2000. PMID: 10975872
-
Dichotomous role of interferon-gamma in allogeneic bone marrow transplant.Biol Blood Marrow Transplant. 2009 Nov;15(11):1347-53. doi: 10.1016/j.bbmt.2009.07.015. Biol Blood Marrow Transplant. 2009. PMID: 19822293 Free PMC article. Review.
-
Unusual patterns of alloimmunity evoked by allogeneic liver parenchymal cells.Immunol Rev. 2000 Apr;174:260-79. doi: 10.1034/j.1600-0528.2002.017409.x. Immunol Rev. 2000. PMID: 10807522 Review.
Cited by
-
Phase II Trial of Costimulation Blockade With Abatacept for Prevention of Acute GVHD.J Clin Oncol. 2021 Jun 10;39(17):1865-1877. doi: 10.1200/JCO.20.01086. Epub 2021 Jan 15. J Clin Oncol. 2021. PMID: 33449816 Free PMC article. Clinical Trial.
-
New strategies for prevention and treatment of graft-versus-host disease and for induction of graft-versus-leukemia effects.Int J Hematol. 2003 Jan;77(1):15-21. doi: 10.1007/BF02982598. Int J Hematol. 2003. PMID: 12568295 Review.
-
α-1-Antitrypsin (AAT)-modified donor cells suppress GVHD but enhance the GVL effect: a role for mitochondrial bioenergetics.Blood. 2014 Oct 30;124(18):2881-91. doi: 10.1182/blood-2014-04-570440. Epub 2014 Sep 15. Blood. 2014. PMID: 25224412 Free PMC article.
-
Immunomodulation of Selective Naive T Cell Functions by p110δ Inactivation Improves the Outcome of Mismatched Cell Transplantation.Cell Rep. 2015 Feb 10;10(5):702-710. doi: 10.1016/j.celrep.2015.01.002. Epub 2015 Feb 5. Cell Rep. 2015. PMID: 25660021 Free PMC article.
-
Immunomodulatory Therapies for the Treatment of Graft-versus-host Disease.Hemasphere. 2021 Jun 1;5(6):e581. doi: 10.1097/HS9.0000000000000581. eCollection 2021 Jun. Hemasphere. 2021. PMID: 34095764 Free PMC article. Review.
References
-
- Blazar BR, Taylor PA, Linsley PS, et al. In vivo blockade of CD28/CTLA4: B7/BB1 interaction with CTLA4-Ig reduces lethal murine graft-versus-host disease across the major histocompatibility complex barrier in mice. Blood. 1994;83:3815–25. - PubMed
-
- Wallace PM, Johnson JS, MacMaster JF, et al. CTLA4Ig treatment ameliorates the lethality of murine graft-versus-host disease across major histocompatibility complex barriers. Transplantation. 1994;58:602–10. - PubMed
-
- Saito K, Yagita H, Hashimoto H, et al. Effect of CD80 and CD86 blockade and anti- interleukin-12 treatment on mouse acute graft-versus-host disease. Eur J Immunol. 1996;26:3098–106. - PubMed
-
- Blazar BR, Taylor PA, Panoskaltsis-Mortari A, et al. Blockade of CD40 ligand–CD40 interaction impairs CD4+ T cell-mediated alloreactivity by inhibiting mature donor T cell expansion and function after bone marrow transplantation. J Immunol. 1997;158:29–39. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

