Testis-specific cytochrome c-null mice produce functional sperm but undergo early testicular atrophy
- PMID: 12101247
- PMCID: PMC133957
- DOI: 10.1128/MCB.22.15.5554-5562.2002
Testis-specific cytochrome c-null mice produce functional sperm but undergo early testicular atrophy
Abstract
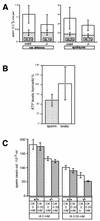
Differentiating male germ cells express a testis-specific form of cytochrome c (Cyt c(T)) that is distinct from the cytochrome c expressed in somatic cells (Cyt c(S)). To examine the role of Cyt c(T) in germ cells, we generated mice null for Cyt c(T). Homozygous Cyt c(T)(-/-) pups were statistically underrepresented (21%) but developed normally and were fertile. However, spermatozoa isolated from the cauda epididymis of Cyt c(T)-null animals were less effective in fertilizing oocytes in vitro and contain reduced levels of ATP compared to wild-type sperm. Sperm from Cyt c(T)-null mice contained a greater number of immotile spermatozoa than did samples from control mice, i.e., 53.1% +/- 13.7% versus 33.2% +/- 10.3% (P < 0.0001) for vas deferens sperm and 40.1% +/- 9.6% versus 33.2% +/- 7.5% (P = 0.0104) for epididymal sperm. Cyt c(T)-null mice often exhibit early atrophy of the testes after 4 months of age, losing germ cells as a result of increased apoptosis. However, no difference in the activation of caspase-3, -8, or -9 was detected between the Cyt c(T)(-/-) testes and controls. Our data indicate that the Cyt c(T)-null testes undergo early atrophy equivalent to that which occurs during aging as a consequence of a reduction in oxidative phosphorylation.
Figures




Similar articles
-
Long-chain fatty acid triglyceride (TG) metabolism disorder impairs male fertility: a study using adipose triglyceride lipase deficient mice.Mol Hum Reprod. 2017 Jul 1;23(7):452-460. doi: 10.1093/molehr/gax031. Mol Hum Reprod. 2017. PMID: 28510703
-
Fertilizing capacity of rat spermatozoa is correlated with decline in straight-line velocity measured by continuous computer-aided sperm analysis: epididymal rat spermatozoa from the proximal cauda have a greater fertilizing capacity in vitro than those from the distal cauda or vas deferens.J Androl. 1996 Jan-Feb;17(1):50-60. J Androl. 1996. PMID: 8833741
-
HANP1/H1T2, a novel histone H1-like protein involved in nuclear formation and sperm fertility.Mol Cell Biol. 2005 Aug;25(16):7107-19. doi: 10.1128/MCB.25.16.7107-7119.2005. Mol Cell Biol. 2005. PMID: 16055721 Free PMC article.
-
Amelioration of heat stress-induced damage to testes and sperm quality.Theriogenology. 2020 Dec;158:84-96. doi: 10.1016/j.theriogenology.2020.08.034. Epub 2020 Sep 8. Theriogenology. 2020. PMID: 32947064 Review.
-
Development and use of surgical procedures to bypass selected regions of the mammalian epididymis: effects on sperm maturation.J Reprod Fertil Suppl. 1998;53:183-95. J Reprod Fertil Suppl. 1998. PMID: 10645277 Review.
Cited by
-
Aberrant expression of sperm‑specific glycolytic enzymes are associated with poor sperm quality.Mol Med Rep. 2019 Apr;19(4):2471-2478. doi: 10.3892/mmr.2019.9926. Epub 2019 Feb 1. Mol Med Rep. 2019. PMID: 30720094 Free PMC article.
-
Transcriptome research on spermatogenic molecular drive in mammals.Asian J Androl. 2015 Nov-Dec;17(6):961-71. doi: 10.4103/1008-682X.159721. Asian J Androl. 2015. PMID: 26306849 Free PMC article. Review.
-
Diverse functions of cytochrome c in cell death and disease.Cell Death Differ. 2024 Apr;31(4):387-404. doi: 10.1038/s41418-024-01284-8. Epub 2024 Mar 23. Cell Death Differ. 2024. PMID: 38521844 Free PMC article. Review.
-
Apoptotic cell death in disease-Current understanding of the NCCD 2023.Cell Death Differ. 2023 May;30(5):1097-1154. doi: 10.1038/s41418-023-01153-w. Epub 2023 Apr 26. Cell Death Differ. 2023. PMID: 37100955 Free PMC article. Review.
-
Targeted disruption of Slc2a8 (GLUT8) reduces motility and mitochondrial potential of spermatozoa.Mol Membr Biol. 2008 Apr;25(3):224-35. doi: 10.1080/09687680701855405. Mol Membr Biol. 2008. PMID: 18428038 Free PMC article.
References
-
- Bourgeron, T. 2000. Mitochondrial function and male infertility. Probl. Cell Differ. 28:187-210. - PubMed
-
- Cai, L., B. F. Hales, and B. Robaire. 1997. Induction of apoptosis in the germ cells of adult male rats after exposure to cyclophosphamide. Biol. Reprod. 56:1490-1497. - PubMed
-
- Chen, G., J. Cizeau, C. Vande Velde, J. H. Park, G. Bozek, J. Bolton, L. Shi, D. Bubik, and A. Greenberg. 1999. Nix and Nip3 form a subfamily of pro-apoptotic mitochondrial proteins. J. Biol. Chem. 274:7-10. - PubMed
-
- Cummins, J. M., A. M. Jequier, and R. Kan. 1994. Molecular biology of human male infertility: links with aging, mitochondrial genetics, and oxidative stress? Mol. Reprod. Dev. 37:345-362. - PubMed
-
- Furuchi, T., K. Masuko, Y. Nishimune, M. Obinata, and Y. Matsui. 1996. Inhibition of testicular germ cell apoptosis and differentiation in mice misexpressing Bcl-2 in spermatogonia. Development 122:1703-1709. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
