Guanidine hydrochloride inhibits the generation of prion "seeds" but not prion protein aggregation in yeast
- PMID: 12101251
- PMCID: PMC133959
- DOI: 10.1128/MCB.22.15.5593-5605.2002
Guanidine hydrochloride inhibits the generation of prion "seeds" but not prion protein aggregation in yeast
Abstract
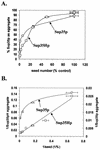
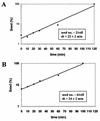
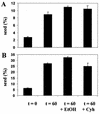
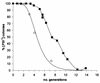
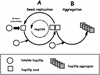
[PSI(+)] strains of the yeast Saccharomyces cerevisiae replicate and transmit the prion form of the Sup35p protein but can be permanently cured of this property when grown in millimolar concentrations of guanidine hydrochloride (GdnHCl). GdnHCl treatment leads to the inhibition of the replication of the [PSI(+)] seeds necessary for continued [PSI(+)] propagation. Here we demonstrate that the rate of incorporation of newly synthesized Sup35p into the high-molecular-weight aggregates, diagnostic of [PSI(+)] strains, is proportional to the number of seeds in the cell, with seed number declining (and the levels of soluble Sup35p increasing) in the presence of GdnHCl. GdnHCl does not cause breakdown of preexisting Sup35p aggregates in [PSI(+)] cells. Transfer of GdnHCl-treated cells to GdnHCl-free medium reverses GdnHCl inhibition of [PSI(+)] seed replication and allows new prion seeds to be generated exponentially in the absence of ongoing protein synthesis. Following such release the [PSI(+)] seed numbers double every 20 to 22 min. Recent evidence (P. C. Ferreira, F. Ness, S. R. Edwards, B. S. Cox, and M. F. Tuite, Mol. Microbiol. 40:1357-1369, 2001; G. Jung and D. C. Masison, Curr. Microbiol. 43:7-10, 2001), together with data presented here, suggests that curing yeast prions by GdnHCl is a consequence of GdnHCl inhibition of the activity of molecular chaperone Hsp104, which in turn is essential for [PSI(+)] propagation. The kinetics of elimination of [PSI(+)] by coexpression of a dominant, ATPase-negative allele of HSP104 were similar to those observed for GdnHCl-induced elimination. Based on these and other data, we propose a two-cycle model for "prionization" of Sup35p in [PSI(+)] cells: cycle A is the GdnHCl-sensitive (Hsp104-dependent) replication of the prion seeds, while cycle B is a GdnHCl-insensitive (Hsp104-independent) process that converts these seeds to pelletable aggregates.
Figures
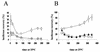






Similar articles
-
The elimination of the yeast [PSI+] prion by guanidine hydrochloride is the result of Hsp104 inactivation.Mol Microbiol. 2001 Jun;40(6):1357-69. doi: 10.1046/j.1365-2958.2001.02478.x. Mol Microbiol. 2001. PMID: 11442834
-
Curing of yeast [PSI+] prion by guanidine inactivation of Hsp104 does not require cell division.Proc Natl Acad Sci U S A. 2005 Sep 6;102(36):12789-94. doi: 10.1073/pnas.0506384102. Epub 2005 Aug 25. Proc Natl Acad Sci U S A. 2005. PMID: 16123122 Free PMC article.
-
Guanidine hydrochloride blocks a critical step in the propagation of the prion-like determinant [PSI(+)] of Saccharomyces cerevisiae.Proc Natl Acad Sci U S A. 2000 Jan 4;97(1):240-4. doi: 10.1073/pnas.97.1.240. Proc Natl Acad Sci U S A. 2000. PMID: 10618402 Free PMC article.
-
The life of [PSI].Curr Genet. 2018 Feb;64(1):1-8. doi: 10.1007/s00294-017-0714-7. Epub 2017 Jun 26. Curr Genet. 2018. PMID: 28653109 Free PMC article. Review.
-
Prion stability.Prion. 2007 Jul-Sep;1(3):170-8. doi: 10.4161/pri.1.3.4839. Epub 2007 Jul 6. Prion. 2007. PMID: 19164897 Free PMC article. Review.
Cited by
-
Influence of prion variant and yeast strain variation on prion-molecular chaperone requirements.Prion. 2011 Oct-Dec;5(4):238-44. doi: 10.4161/pri.17818. Epub 2011 Oct 1. Prion. 2011. PMID: 22156732 Free PMC article.
-
Primary sequence independence for prion formation.Proc Natl Acad Sci U S A. 2005 Sep 6;102(36):12825-30. doi: 10.1073/pnas.0506136102. Epub 2005 Aug 25. Proc Natl Acad Sci U S A. 2005. PMID: 16123127 Free PMC article.
-
Defining the limits: Protein aggregation and toxicity in vivo.Crit Rev Biochem Mol Biol. 2014 Jul-Aug;49(4):294-303. doi: 10.3109/10409238.2014.914151. Epub 2014 Apr 28. Crit Rev Biochem Mol Biol. 2014. PMID: 24766537 Free PMC article. Review.
-
Hsp104 disaggregase at normal levels cures many [PSI+] prion variants in a process promoted by Sti1p, Hsp90, and Sis1p.Proc Natl Acad Sci U S A. 2017 May 23;114(21):E4193-E4202. doi: 10.1073/pnas.1704016114. Epub 2017 May 8. Proc Natl Acad Sci U S A. 2017. PMID: 28484020 Free PMC article.
-
Schizosaccharomyces pombe disaggregation machinery chaperones support Saccharomyces cerevisiae growth and prion propagation.Eukaryot Cell. 2013 May;12(5):739-45. doi: 10.1128/EC.00301-12. Epub 2013 Mar 15. Eukaryot Cell. 2013. PMID: 23504563 Free PMC article.
References
-
- Caughey, B. 2000. Transmissible spongiform encephalopathies, amyloidoses and yeast prions: common threads? Nat. Med. 6:751-754. - PubMed
-
- Caughey, B. 2001. Interactions between prion protein isoforms: the kiss of death. Trends Biochem. Sci. 26:235-242. - PubMed
-
- Chernoff, Y. O., I. L. Derkach, A. Dagkesamanskaya, V. Tikhomironva, M. D. Ter-Avanesyan, and S. Inge-Vechtomov. 1988. Nonsense-suppression by amplification of translational protein factor gene. Dokl. Akad. Nauk SSSR (Biol. Sci.) 301:1227-1229. - PubMed
-
- Chernoff, Y. O., I. L. Derkach, and S. G. Inge-Vechtomov. 1993. Multicopy SUP35 gene induces de-novo appearance of psi-like factors in the yeast Saccharomyces cerevisiae. Curr. Genet. 24:268-270. - PubMed
-
- Chernoff, Y. O., S. L. Lindquist, B.-I. Ono, S. G. Inge-Vechtomov, and S. W. Liebman. 1995. Role of the chaperone protein Hsp104 in propagation of the yeast prion-like factor [psi+]. Science 268:880-884. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
