Phosphorylation by p38MAPK and recruitment of SUG-1 are required for RA-induced RAR gamma degradation and transactivation
- PMID: 12110588
- PMCID: PMC126119
- DOI: 10.1093/emboj/cdf374
Phosphorylation by p38MAPK and recruitment of SUG-1 are required for RA-induced RAR gamma degradation and transactivation
Abstract
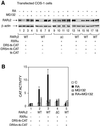
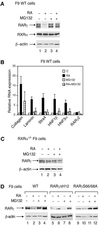
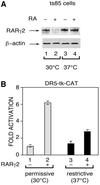
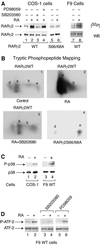
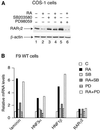
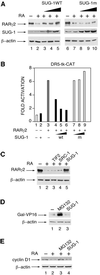
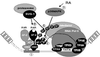
The nuclear retinoic acid receptor RAR gamma 2 undergoes proteasome-dependent degradation upon ligand binding. Here we provide evidence that the domains that signal proteasome-mediated degradation overlap with those that activate transcription, i.e. the activation domains AF-1 and AF-2. The AF-1 domain signals RAR gamma 2 degradation through its phosphorylation by p38MAPK in response to RA. The AF-2 domain acts via the recruitment of SUG-1, which belongs to the 19S regulatory subunit of the 26S proteasome. Blocking RAR gamma 2 degradation through inhibition of either the p38MAPK pathway or the 26S proteasome function impairs its RA-induced transactivation activity. Thus, the turnover of RAR gamma 2 is linked to transactivation.
Figures

References
-
- Alsayed Y., Uddin,S., Mahmud,N., Lekmine,F., Kalvakolanu,D.V., Minucci,S., Bokoch,G. and Platanias,L.C. (2001) Activation of Rac1 and the p38 mitogen-activated protein kinase pathway in response to all-trans-retinoic acid. J. Biol. Chem., 276, 4012–4019. - PubMed
-
- Bastien J., Adam-Stitah,S., Riedl,T., Egly,J.M., Chambon,P. and Rochette-Egly,C. (2000) TFIIH interacts with the retinoic acid receptor γ and phosphorylates its AF-1-activating domain through cdk7. J. Biol. Chem., 275, 21896–21904. - PubMed
-
- Boyle J.O., Langenfeld,J., Lonardo,F., Sekula,D., Reczek,P., Rusch,V., Dawson,M.I., Dmitrovsky,E. (1999) Cyclin D1 proteolysis: a retinoid chemoprevention signal in normal, immortalized and transformed human bronchial epithelial cells. J. Natl Cancer Inst., 91, 373–379. - PubMed
-
- Chambon P. (1996) A decade of molecular biology on retinoic acid receptors. FASEB J., 10, 940–954. - PubMed
-
- Chen J.D. (2000) Steroid/nuclear receptor coactivators. Vitam. Horm., 58, 391–448. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

