Global repression of exotoxin synthesis by staphylococcal superantigens
- PMID: 12110733
- PMCID: PMC126631
- DOI: 10.1073/pnas.152152499
Global repression of exotoxin synthesis by staphylococcal superantigens
Abstract
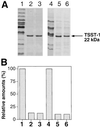
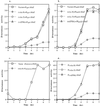
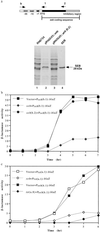
Virulent Staphylococcus aureus strains typically produce and secrete large quantities of many extracellular proteins involved in pathogenesis. Such strains cause the classical staphylococcal lesion--local tissue destruction and aggressive inflammation accompanied by the massive influx of polymorphonuclear leukocytes, leading to the formation of pus. Most strains causing toxic shock syndrome, however, produce and secrete very small quantities of most exoproteins although they elaborate high levels of toxic shock syndrome toxin-1 (TSST-1). These strains cause local infections that are remarkably apurulent although potentially fatal owing to the superantigen. We have analyzed this disparity and have found that TSST-1 itself is a negative global regulator of exoprotein gene transcription. TSST-1 not only represses most exoprotein genes but determines its own high expression level by autorepression. We report also that a second superantigen, enterotoxin B, has similar regulatory properties.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

