Up-regulation of Fas (CD95) and induction of apoptosis in intestinal epithelial cells by nematode-derived molecules
- PMID: 12117905
- PMCID: PMC128120
- DOI: 10.1128/IAI.70.8.4002-4008.2002
Up-regulation of Fas (CD95) and induction of apoptosis in intestinal epithelial cells by nematode-derived molecules
Abstract
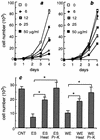
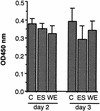
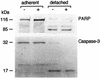
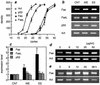
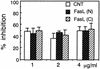
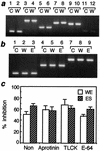
Infection by the intestinal nematode Nippostrongylus brasiliensis induces acceleration of apoptosis in the small intestinal villus epithelial cells in vivo. In the present study, we examined whether worm extract or excretory-secretory product induces apoptosis in the rat intestinal epithelial cell line IEC-6 in vitro. In the presence of worm extract or excretory-secretory product (> or =6 microg/ml), IEC-6 cell growth was significantly suppressed, and there was a concomitant increase in the number of detached cells in culture dishes. Detached cells showed nuclear fragmentation, activation of caspase-3, and specific cleavage of poly(ADP-ribose) polymerase, suggesting that apoptosis was induced in these cells. Semiquantitative reverse transcription-PCR showed that expression of Fas (CD95) mRNA was up-regulated as early as 6 h after addition of excretory-secretory product, while Fas ligand expression and p53 expression were not up-regulated. Fluorescence-activated cell sorter analyses revealed a significant increase in Fas expression and a slight increase in FasL expression in IEC-6 cells cultured in the presence of excretory-secretory product, while control IEC-6 cells expressed neither Fas or FasL. These results indicated that N. brasiliensis worms produce and secrete biologically active molecules that trigger apoptosis in intestinal epithelial cells together with up-regulation of Fas expression, although the mechanism of induction of apoptosis remains to be elucidated.
Figures

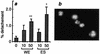

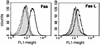
Similar articles
-
Interleukin-11 antagonizes Fas ligand-mediated apoptosis in IEC-18 intestinal epithelial crypt cells: role of MEK and Akt-dependent signaling.Am J Physiol Gastrointest Liver Physiol. 2008 Mar;294(3):G728-37. doi: 10.1152/ajpgi.00002.2007. Epub 2008 Jan 17. Am J Physiol Gastrointest Liver Physiol. 2008. PMID: 18202115
-
Activation of caspases in intestinal villus epithelial cells of normal and nematode infected rats.Gut. 2002 Jan;50(1):71-7. doi: 10.1136/gut.50.1.71. Gut. 2002. PMID: 11772970 Free PMC article.
-
Fas ligand- mediated killing by intestinal intraepithelial lymphocytes. Participation in intestinal graft-versus-host disease.J Clin Invest. 1998 Feb 1;101(3):570-7. doi: 10.1172/JCI896. J Clin Invest. 1998. PMID: 9449689 Free PMC article.
-
Direct effects of IL-4/IL-13 and the nematode Nippostrongylus brasiliensis on intestinal epithelial cells in vitro.Parasite Immunol. 2010 Jun;32(6):420-9. doi: 10.1111/j.1365-3024.2010.01200.x. Parasite Immunol. 2010. PMID: 20500673
-
Sustained polymorphonuclear leukocyte transmigration induces apoptosis in T84 intestinal epithelial cells.J Cell Biol. 2000 Sep 18;150(6):1479-88. doi: 10.1083/jcb.150.6.1479. J Cell Biol. 2000. PMID: 10995451 Free PMC article.
Cited by
-
The influence of selected gastrointestinal parasites on apoptosis in intestinal epithelial cells.Biomolecules. 2020 Apr 27;10(5):674. doi: 10.3390/biom10050674. Biomolecules. 2020. PMID: 32349424 Free PMC article. Review.
-
Hymenolepis diminuta Infection Affects Apoptosis in the Small and Large Intestine.Int J Environ Res Public Health. 2022 Aug 8;19(15):9753. doi: 10.3390/ijerph19159753. Int J Environ Res Public Health. 2022. PMID: 35955110 Free PMC article.
-
The occupation of intestinal epithelium by Trichinella spiralis in BALB/C mice is not associated with local manifestation of apoptosis related factors.Parasitol Res. 2013 Nov;112(11):3917-24. doi: 10.1007/s00436-013-3582-1. Epub 2013 Sep 15. Parasitol Res. 2013. PMID: 24037538
References
-
- Appella, E., and C. W. Anderson. 2000. Signaling to p53: breaking the posttranslational modification code. Pathol. Biol. 48:227-245. - PubMed
-
- Batra, S., S. P. Singh, S. Gupta, J. C. Katiyar, and V. M. Srivastava. 1990. Reactive oxygen intermediates metabolizing enzymes in Ancylostoma ceylanicum and Nippostrongylus brasiliensis. Free Radical Biol. Med. 8:271-274. - PubMed
-
- Burt, J. S., and B. M. Ogilvie. 1975. In vitro maintenance of nematode parasites assayed by acetylcholinesterase and allergen secretion. Exp. Parasitol. 38:75-82. - PubMed
-
- Chen, X. M., G. J. Gores, C. V. Paya, and N. F. LaRusso. 1999. Cryptosporidium parvum induces apoptosis in biliary epithelia by a Fas/Fas ligand-dependent mechanism. Am. J. Physiol. 277:g599-608. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

