Roles of p38 mitogen-activated protein kinase, NF-kappaB, and protein kinase C in proinflammatory cytokine mRNA expression by human peripheral blood leukocytes, monocytes, and neutrophils in response to Anaplasma phagocytophila
- PMID: 12117921
- PMCID: PMC128199
- DOI: 10.1128/IAI.70.8.4132-4141.2002
Roles of p38 mitogen-activated protein kinase, NF-kappaB, and protein kinase C in proinflammatory cytokine mRNA expression by human peripheral blood leukocytes, monocytes, and neutrophils in response to Anaplasma phagocytophila
Abstract
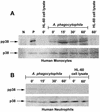
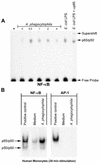
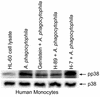
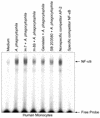
Anaplasma phagocytophila, an obligately intracellular bacterium of granulocytes, causes human granulocytic ehrlichiosis. Within 2 h after addition of A. phagocytophila, interleukin-1beta (IL-1beta), tumor necrosis factor alpha (TNF-alpha), and IL-6 mRNAs are induced in human peripheral blood leukocytes (PBLs) or monocytes in vitro. However, neutrophils generate only IL-1beta mRNA. In the present study, signaling pathways for induction of these three cytokines were examined. TNF-alpha and IL-6 mRNA expression by PBLs was inhibited with SB 203580 (a p38 mitogen-activated protein kinase [MAPK] inhibitor), MG-132 (a proteasome inhibitor), and SN-50 (an NF-kappaB inhibitor). Activation of p38 MAPK and NF-kappaB mRNAs in monocytes was detectable within 15 to 30 min after addition of A. phagocytophila. Expression of these two cytokine mRNAs in PBLs and monocytes was also dependent on protein kinase C (PKC), protein kinase A (PKA), and protein tyrosine kinase (PTK). IL-1beta mRNA expression by neutrophils was not dependent on p38 MAPK, and p38 MAPK was not activated in neutrophils incubated with A. phagocytophila. IL-1beta mRNA induction by PBLs, monocytes, and neutrophils was dependent on PKC and PKA. Neutrophil expression of IL-1beta mRNA was dependent on transglutaminase, phospholipase C, and PTK, all of which are also required for internalization of A. phagocytophila. However, monocyte expression of IL-1beta mRNA was less dependent on these enzymes. These results suggest that A. phagocytophila transduces different signals between its host neutrophils and monocytes for proinflammatory cytokine generation.
Figures



Similar articles
-
Expression of interleukin-1beta, tumor necrosis factor alpha, and interleukin-6 in human peripheral blood leukocytes exposed to human granulocytic ehrlichiosis agent or recombinant major surface protein P44.Infect Immun. 2000 Jun;68(6):3394-402. doi: 10.1128/IAI.68.6.3394-3402.2000. Infect Immun. 2000. PMID: 10816490 Free PMC article.
-
TNF-alpha, inefficient by itself, potentiates IL-1beta-induced PGHS-2 expression in human pulmonary microvascular endothelial cells: requirement of NF-kappaB and p38 MAPK pathways.Br J Pharmacol. 2002 Aug;136(7):1005-14. doi: 10.1038/sj.bjp.0704811. Br J Pharmacol. 2002. PMID: 12145100 Free PMC article.
-
A Mycoplasma fermentans-derived synthetic lipopeptide induces AP-1 and NF-kappaB activity and cytokine secretion in macrophages via the activation of mitogen-activated protein kinase pathways.J Biol Chem. 1998 Dec 18;273(51):34391-8. doi: 10.1074/jbc.273.51.34391. J Biol Chem. 1998. PMID: 9852105
-
Anti-Ehrlichia chaffeensis antibody complexed with E. chaffeensis induces potent proinflammatory cytokine mRNA expression in human monocytes through sustained reduction of IkappaB-alpha and activation of NF-kappaB.Infect Immun. 1997 Jul;65(7):2890-7. doi: 10.1128/iai.65.7.2890-2897.1997. Infect Immun. 1997. PMID: 9199464 Free PMC article.
-
Proinflammatory cytokines, IL-1β and TNF-α, induce expression of interleukin-34 mRNA via JNK- and p44/42 MAPK-NF-κB pathway but not p38 pathway in osteoblasts.Rheumatol Int. 2011 Nov;31(11):1525-30. doi: 10.1007/s00296-010-1688-7. Epub 2010 Dec 23. Rheumatol Int. 2011. PMID: 21181166
Cited by
-
Transcriptome analysis of the hepatopancreas from the Litopenaeus vannamei infected with different flagellum types of Vibrio alginolyticus strains.Front Cell Infect Microbiol. 2023 Nov 21;13:1265917. doi: 10.3389/fcimb.2023.1265917. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 38076457 Free PMC article.
-
Sea buckthorn flavonoids and their derivatives: potential natural compounds for the treatment of diabetic cardiomyopathy.Front Pharmacol. 2025 Jul 17;16:1599756. doi: 10.3389/fphar.2025.1599756. eCollection 2025. Front Pharmacol. 2025. PMID: 40746722 Free PMC article. Review.
-
Early transcriptional response of human neutrophils to Anaplasma phagocytophilum infection.Infect Immun. 2005 Dec;73(12):8089-99. doi: 10.1128/IAI.73.12.8089-8099.2005. Infect Immun. 2005. PMID: 16299303 Free PMC article.
-
Human Granulocytic Anaplasmosis as a COVID-19 Mimicker.Eur J Case Rep Intern Med. 2021 Dec 6;8(12):003047. doi: 10.12890/2021_003047. eCollection 2021. Eur J Case Rep Intern Med. 2021. PMID: 35059343 Free PMC article.
-
Functional inhibition or genetic deletion of acid sphingomyelinase bacteriostatically inhibits Anaplasma phagocytophilum infection in vivo.Pathog Dis. 2021 Jan 6;79(1):ftaa072. doi: 10.1093/femspd/ftaa072. Pathog Dis. 2021. PMID: 33220685 Free PMC article.
References
-
- Akiyama, T., and H. Ogawara. 1991. Use and specificity of genistein as inhibitor of protein-tyrosine kinases. Methods Enzymol. 201:362-370. - PubMed
-
- Alessi, D. R., A. Cuenda, P. Cohen, D. T. Dudley, and A. R. Saltiel. 1995. PD 098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J. Biol. Chem. 270:27489-27494. - PubMed
-
- Bakken, J. S., J. Krueth, C. Wilson-Nordskog, R. L. Tilden, K. Asanovich, and J. S. Dumler. 1996. Clinical and laboratory characteristics of human granulocytic ehrlichiosis. JAMA 275:199-205. - PubMed
-
- Bakken, J. S., J. Krueth, R. L. Tilden, J. S. Dumler, and R. E. Kristansen. 1996. Serologic evidence of human granulocytic ehrlichiosis in Norway. Eur. J. Clin. Microbiol. Infect. Dis. 15:829-832. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

