Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death
- PMID: 12121617
- PMCID: PMC4030438
- DOI: 10.1016/s0960-9822(02)00904-1
Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death
Abstract
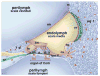
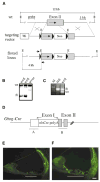
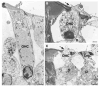
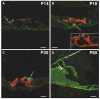
Mutations in the gene encoding the gap junction protein connexin26 (Cx26) are responsible for the autosomal recessive isolated deafness, DFNB1, which accounts for half of the cases of prelingual profound hereditary deafness in Caucasian populations. To date, in vivo approaches to decipher the role of Cx26 in the inner ear have been hampered by the embryonic lethality of the Cx26 knockout mice. To overcome this difficulty, we performed targeted ablation of Cx26 specifically in one of the two cellular networks that it underlies in the inner ear, namely, the epithelial network. We show that homozygous mutant mice, Cx26(OtogCre), have hearing impairment, but no vestibular dysfunction. The inner ear developed normally. However, on postnatal day 14 (P14), i.e., soon after the onset of hearing, cell death appeared and eventually extended to the cochlear epithelial network and sensory hair cells. Cell death initially affected only the supporting cells of the genuine sensory cell (inner hair cell, IHC), thus suggesting that it could be triggered by the IHC response to sound stimulation. Altogether, our results demonstrate that the Cx26-containing epithelial gap junction network is essential for cochlear function and cell survival. We conclude that prevention of cell death in the sensory epithelium is essential for any attempt to restore the auditory function in DFNB1 patients.
Figures

Similar articles
-
Gap junctions and connexin expression in the inner ear.Novartis Found Symp. 1999;219:134-50; discussion 151-6. doi: 10.1002/9780470515587.ch9. Novartis Found Symp. 1999. PMID: 10207902 Review.
-
A deafness mechanism of digenic Cx26 (GJB2) and Cx30 (GJB6) mutations: Reduction of endocochlear potential by impairment of heterogeneous gap junctional function in the cochlear lateral wall.Neurobiol Dis. 2017 Dec;108:195-203. doi: 10.1016/j.nbd.2017.08.002. Epub 2017 Aug 17. Neurobiol Dis. 2017. PMID: 28823936 Free PMC article.
-
Mice with conditional deletion of Cx26 exhibit no vestibular phenotype despite secondary loss of Cx30 in the vestibular end organs.Hear Res. 2015 Oct;328:102-12. doi: 10.1016/j.heares.2015.07.018. Epub 2015 Jul 29. Hear Res. 2015. PMID: 26232528 Free PMC article.
-
Connexins and gap junctions in the inner ear--it's not just about K⁺ recycling.Cell Tissue Res. 2015 Jun;360(3):633-44. doi: 10.1007/s00441-014-2029-z. Epub 2014 Nov 9. Cell Tissue Res. 2015. PMID: 25381570 Free PMC article. Review.
-
Early developmental expression of connexin26 in the cochlea contributes to its dominate functional role in the cochlear gap junctions.Biochem Biophys Res Commun. 2012 Jan 6;417(1):245-50. doi: 10.1016/j.bbrc.2011.11.093. Epub 2011 Nov 28. Biochem Biophys Res Commun. 2012. PMID: 22142852 Free PMC article.
Cited by
-
Phenotypic variability in gap junction syndromic skin disorders: experience from KID and Clouston syndromes' clinical diagnostics.J Appl Genet. 2015 Aug;56(3):329-37. doi: 10.1007/s13353-014-0266-1. Epub 2015 Jan 10. J Appl Genet. 2015. PMID: 25575739 Free PMC article.
-
Spontaneous and Acetylcholine Evoked Calcium Transients in the Developing Mouse Utricle.Front Cell Neurosci. 2019 May 7;13:186. doi: 10.3389/fncel.2019.00186. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31133810 Free PMC article.
-
Early Hearing Loss upon Disruption of Slc4a10 in C57BL/6 Mice.J Assoc Res Otolaryngol. 2019 Jun;20(3):233-245. doi: 10.1007/s10162-019-00719-1. Epub 2019 Apr 18. J Assoc Res Otolaryngol. 2019. PMID: 31001720 Free PMC article.
-
How do taste cells lacking synapses mediate neurotransmission? CALHM1, a voltage-gated ATP channel.Bioessays. 2013 Dec;35(12):1111-8. doi: 10.1002/bies.201300077. Epub 2013 Sep 17. Bioessays. 2013. PMID: 24105910 Free PMC article. Review.
-
The endocochlear potential as an indicator of reticular lamina integrity after noise exposure in mice.Hear Res. 2018 Apr;361:138-151. doi: 10.1016/j.heares.2018.01.015. Epub 2018 Feb 1. Hear Res. 2018. PMID: 29426600 Free PMC article.
References
-
- Kelsell DP, Dunlop J, Stevens HP, Lench NJ, Liang JN, Parry G, Mueller RF, Leigh IM. Connexin 26 mutations in hereditary non-syndromic sensorineural deafness. Nature. 1997;387:80–83. - PubMed
-
- Denoyelle F, Weil D, Maw MA, Wilcox SA, Lench NJ, Allen-Powell DR, Osborn AH, Dahl HHM, Middleton A, Houseman MJ, et al. Prelingual deafness: high prevalence of a 30delG mutation in the connexin 26 gene. Hum Mol Genet. 1997;6:2173–2177. - PubMed
-
- Zelante L, Gasparini P, Estivill X, Melchionda S, D’Agruma L, Govea N, Mila M, Della Monica M, Lutfi J, Shohat M, et al. Connexin26 mutations associated with the most common form of non-syndromic neurosensory autosomal recessive deafness (DFNB1) in Mediterraneans. Hum Mol Genet. 1997;6:1605–1609. - PubMed
-
- Rabionet R, Gasparini P, Estivill X. Molecular genetics of hearing impairment due to mutations in gap junction genes encoding β-connexins. Hum Mutat. 2000;16:190–202. - PubMed
-
- Petit C, Levilliers J, Hardelin JP. Molecular genetics of hearing loss. Annu Rev Genet. 2001;35:859–865. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

