Emp47p and its close homolog Emp46p have a tyrosine-containing endoplasmic reticulum exit signal and function in glycoprotein secretion in Saccharomyces cerevisiae
- PMID: 12134087
- PMCID: PMC117331
- DOI: 10.1091/mbc.e02-01-0027
Emp47p and its close homolog Emp46p have a tyrosine-containing endoplasmic reticulum exit signal and function in glycoprotein secretion in Saccharomyces cerevisiae
Abstract
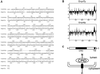
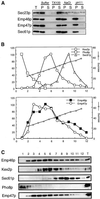
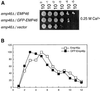
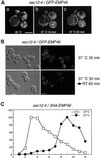
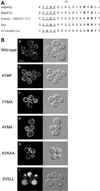
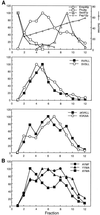
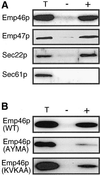
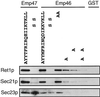
The yeast open reading frame YLR080w/EMP46 encodes a homolog of the Golgi protein Emp47p. These two proteins are 45% identical and have a single transmembrane domain in their C-terminal regions and a carbohydrate recognition domain signature in the N-terminal region. The C-terminal tail of Emp46p includes a dilysine signal. This protein is localized to Golgi membranes at steady state by subcellular fractionation and green fluorescent protein labeling. On block of forward transport in sec12-4 cells, redistribution of Emp46p from the Golgi to the endoplasmic reticulum is observed. These localization features are similar to those previously reported for Emp47p. In addition, mutagenesis of the C-terminal region identified a tyrosine-containing motif as a critical determinant of the Golgi-localization and interaction with both COPI and COPII components. Similar motifs are also observed in the C-terminal tail of Emp47p and other mammalian homologs. Disruption of Emp47p displays a growth defect at a high temperature or on Ca(2+)-containing medium, which is rescued by overexpression of Emp46p, suggesting a partially overlapping function between Emp46p and Emp47p. In addition, we found that the disruption of both Emp46p and Emp47p show a marked defect in the secretion of a subset of glycoproteins. Analysis of the C-terminal mutants for Ca(2+) sensitivity revealed that the forward transport of Emp46/47p is essential for their function, whereas the retrograde transport is not. We propose that Emp46p and Emp47p are required for the export of specific glycoprotein cargo from the endoplasmic reticulum.
Figures




Similar articles
-
Structures of the carbohydrate recognition domain of Ca2+-independent cargo receptors Emp46p and Emp47p.J Biol Chem. 2006 Apr 14;281(15):10410-9. doi: 10.1074/jbc.M512258200. Epub 2006 Jan 26. J Biol Chem. 2006. PMID: 16439369
-
Oligomerization of a cargo receptor directs protein sorting into COPII-coated transport vesicles.Mol Biol Cell. 2003 Jul;14(7):3055-63. doi: 10.1091/mbc.e03-02-0115. Epub 2003 Apr 17. Mol Biol Cell. 2003. PMID: 12857885 Free PMC article.
-
The Golgi-localization of yeast Emp47p depends on its di-lysine motif but is not affected by the ret1-1 mutation in alpha-COP.J Cell Biol. 1995 Nov;131(4):895-912. doi: 10.1083/jcb.131.4.895. J Cell Biol. 1995. PMID: 7490292 Free PMC article.
-
Making COPII coats.Cell. 2007 Jun 29;129(7):1251-2. doi: 10.1016/j.cell.2007.06.015. Cell. 2007. PMID: 17604713 Review.
-
Sorting and Export of Proteins at the Endoplasmic Reticulum.Cold Spring Harb Perspect Biol. 2023 May 2;15(5):a041258. doi: 10.1101/cshperspect.a041258. Cold Spring Harb Perspect Biol. 2023. PMID: 35940902 Free PMC article. Review.
Cited by
-
Modulating endoplasmic reticulum-Golgi cargo receptors for improving secretion of carrier-fused heterologous proteins in the filamentous fungus Aspergillus oryzae.Appl Environ Microbiol. 2015 Jan;81(2):533-43. doi: 10.1128/AEM.02133-14. Epub 2014 Oct 31. Appl Environ Microbiol. 2015. PMID: 25362068 Free PMC article.
-
A novel endoplasmic reticulum export signal: proline at the +2-position from the signal peptide cleavage site.J Biol Chem. 2009 Oct 2;284(40):27500-10. doi: 10.1074/jbc.M109.021592. Epub 2009 Aug 5. J Biol Chem. 2009. PMID: 19656946 Free PMC article.
-
Legionella pneumophila LegC7 effector protein drives aberrant endoplasmic reticulum:endosome contacts in yeast.Traffic. 2021 Aug;22(8):284-302. doi: 10.1111/tra.12807. Epub 2021 Jul 7. Traffic. 2021. PMID: 34184807 Free PMC article.
-
Maturation-driven transport and AP-1-dependent recycling of a secretory cargo in the Golgi.J Cell Biol. 2019 May 6;218(5):1582-1601. doi: 10.1083/jcb.201807195. Epub 2019 Mar 11. J Cell Biol. 2019. PMID: 30858194 Free PMC article.
-
Two novel WD40 domain-containing proteins, Ere1 and Ere2, function in the retromer-mediated endosomal recycling pathway.Mol Biol Cell. 2011 Nov;22(21):4093-107. doi: 10.1091/mbc.E11-05-0440. Epub 2011 Aug 31. Mol Biol Cell. 2011. PMID: 21880895 Free PMC article.
References
-
- Appenzeller C, Andersson H, Kappeler F, Hauri HP. The lectin ERGIC-53 is a cargo transport receptor for glycoproteins. Nat Cell Biol. 1999;1:330–334. - PubMed
-
- Barlowe C, Orci L, Yeung T, Hosobuchi M, Hamamoto S, Salama N, Rexach MF, Ravazzola M, Amherdt M, Schekman R. COPII: a membrane coat formed by Sec proteins that drives vesicle budding from the endoplasmic reticulum. Cell. 1994;77:895–907. - PubMed
-
- Barlowe C, Schekman R. SEC12 encodes a guanine-nucleotide-exchange factor essential for transport vesicle budding from the ER. Nature. 1993;365:347–349. - PubMed
-
- Bednarek SY, Ravazzola M, Hosobuchi M, Amherdt M, Perrelet A, Schekman R, Orci L. COPI- and COPII-coated vesicles bud directly from the endoplasmic reticulum in yeast. Cell. 1995;83:1183–1196. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

