Steady-state nuclear localization of exportin-t involves RanGTP binding and two distinct nuclear pore complex interaction domains
- PMID: 12138183
- PMCID: PMC133969
- DOI: 10.1128/MCB.22.16.5708-5720.2002
Steady-state nuclear localization of exportin-t involves RanGTP binding and two distinct nuclear pore complex interaction domains
Abstract
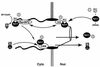
Vertebrate tRNA export receptor exportin-t (Xpo-t) binds to RanGTP and mature tRNAs cooperatively to form a nuclear export complex. Xpo-t shuttles bidirectionally through nuclear pore complexes (NPCs) but is mainly nuclear at steady state. The steady-state distribution of Xpo-t is shown to depend on its interaction with RanGTP. Two distinct Xpo-t NPC interaction domains that bind differentially to peripherally localized nucleoporins in vitro are identified. The N terminus binds to both Nup153 and RanBP2/Nup358 in a RanGTP-dependent manner, while the C terminus binds to CAN/Nup214 independently of Ran. We propose that these interactions increase the concentration of tRNA export complexes and of empty Xpo-t in the vicinity of NPCs and thus increase the efficiency of the Xpo-t transport cycle.
Figures

References
-
- Allen, N. P. C., L. Huang, A. Burlingame, and M. Rexach. 2001. Proteomic analysis of nucleoporin interacting proteins. J. Biol. Chem. 276:29268-29274. - PubMed
-
- Arts, G.-J., M. Fornerod, and I. W. Mattaj. 1998. Identification of a nuclear export receptor for tRNA. Curr. Biol. 8:305-314. - PubMed
-
- Bayliss, R., T. Littlewood, and M. Stewart. 2000. Structural basis for the interaction between FxFG nucleoporin repeats and importin-beta in nuclear trafficking. Cell 102:99-108. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
