Mos is not required for the initiation of meiotic maturation in Xenopus oocytes
- PMID: 12145203
- PMCID: PMC126146
- DOI: 10.1093/emboj/cdf400
Mos is not required for the initiation of meiotic maturation in Xenopus oocytes
Abstract
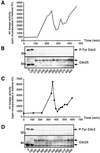
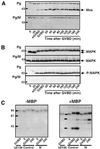
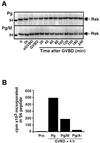
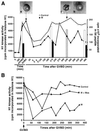
In Xenopus oocytes, the c-mos proto-oncogene product has been proposed to act downstream of progesterone to control the entry into meiosis I, the transition from meiosis I to meiosis II, which is characterized by the absence of S phase, and the metaphase II arrest seen prior to fertilization. Here, we report that inhibition of Mos synthesis by morpholino antisense oligonucleotides does not prevent the progesterone-induced initiation of Xenopus oocyte meiotic maturation, as previously thought. Mos-depleted oocytes complete meiosis I but fail to arrest at metaphase II, entering a series of embryonic-like cell cycles accompanied by oscillations of Cdc2 activity and DNA replication. We propose that the unique and conserved role of Mos is to prevent mitotic cell cycles of the female gamete until the fertilization in Xenopus, starfish and mouse oocytes.
Figures


Similar articles
-
Redundant pathways for Cdc2 activation in Xenopus oocyte: either cyclin B or Mos synthesis.EMBO Rep. 2006 Mar;7(3):321-5. doi: 10.1038/sj.embor.7400611. Epub 2005 Dec 16. EMBO Rep. 2006. PMID: 16374506 Free PMC article.
-
Molecular mechanisms of meiotic maturation and arrest in fish and amphibian oocytes.Semin Cell Dev Biol. 1998 Oct;9(5):569-79. doi: 10.1006/scdb.1998.0251. Semin Cell Dev Biol. 1998. PMID: 9835645 Review.
-
Polo-like kinase confers MPF autoamplification competence to growing Xenopus oocytes.Development. 2004 Apr;131(7):1543-52. doi: 10.1242/dev.01050. Epub 2004 Feb 25. Development. 2004. PMID: 14985258
-
Suppression of DNA replication via Mos function during meiotic divisions in Xenopus oocytes.EMBO J. 1994 May 15;13(10):2399-410. doi: 10.1002/j.1460-2075.1994.tb06524.x. EMBO J. 1994. PMID: 8194530 Free PMC article.
-
Synthesis and function of Mos: the control switch of vertebrate oocyte meiosis.Bioessays. 1997 Jan;19(1):23-8. doi: 10.1002/bies.950190106. Bioessays. 1997. PMID: 9008414 Review.
Cited by
-
Enforcing temporal control of maternal mRNA translation during oocyte cell-cycle progression.EMBO J. 2010 Jan 20;29(2):387-97. doi: 10.1038/emboj.2009.337. Epub 2009 Dec 3. EMBO J. 2010. PMID: 19959990 Free PMC article.
-
The phosphorylation of ARPP19 by Greatwall renders the auto-amplification of MPF independently of PKA in Xenopus oocytes.J Cell Sci. 2013 Sep 1;126(Pt 17):3916-26. doi: 10.1242/jcs.126599. Epub 2013 Jun 18. J Cell Sci. 2013. PMID: 23781026 Free PMC article.
-
Mechanistic studies of the mitotic activation of Mos.Mol Cell Biol. 2006 Jul;26(14):5300-9. doi: 10.1128/MCB.00273-06. Mol Cell Biol. 2006. PMID: 16809767 Free PMC article.
-
MPF-based meiotic cell cycle control: Half a century of lessons from starfish oocytes.Proc Jpn Acad Ser B Phys Biol Sci. 2018;94(4):180-203. doi: 10.2183/pjab.94.013. Proc Jpn Acad Ser B Phys Biol Sci. 2018. PMID: 29643273 Free PMC article. Review.
-
Xp38gamma/SAPK3 promotes meiotic G(2)/M transition in Xenopus oocytes and activates Cdc25C.EMBO J. 2003 Nov 3;22(21):5746-56. doi: 10.1093/emboj/cdg559. EMBO J. 2003. PMID: 14592973 Free PMC article.
References
-
- Bhatt R.R. and Ferrell,J.E.,Jr (1999) The protein kinase p90rsk as an essential mediator of cytostatic factor activity. Science, 286, 1362–1365. - PubMed
-
- Choi T., Rulong,S., Resau,J., Fukasawa,K., Matten,W., Kuriyama,R., Mansour,S., Ahn,N. and Van de Woude,G.F. (1996) Mos/mitogen-activated protein kinase can induce early meiotic phenotypes in the absence of maturation-promoting factor—a novel system for analyzing spindle formation during meiosis I. Proc. Natl Acad. Sci. USA, 93, 4730–4735. - PMC - PubMed
-
- Colledge W.H., Carlton,M.B.L., Udy,G.B. and Evans,M.J. (1994) Disruption of c-mos causes parthenogenetic development of unfertilized mouse eggs. Nature, 370, 65–68. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

