Inactivation of phosphoglycerate mutase and creatine kinase isoenzymes in human serum
- PMID: 12147715
- PMCID: PMC1187187
- DOI: 10.1136/mp.55.4.242
Inactivation of phosphoglycerate mutase and creatine kinase isoenzymes in human serum
Abstract
Aims/background: Total phosphoglycerate mutase (PGM) activity in serum has been shown to be increased in acute myocardial infarction with the same time course as creatine kinase (CK) activity. However, the increase in the muscle (MM) and in the cardiac (MB) PGM isoenzymes was not as high as expected. The present study was undertaken to characterise PGM inactivation by serum and to compare it with serum CK inactivation.
Methods: The PGM and the CK activities of extracts of human heart, skeletal muscle, and brain were determined spectrophotometrically after incubation with different media, namely: plasma, whole serum, dialysed serum, heated serum, serum ultrafiltrate, urate solution, and buffer solution.
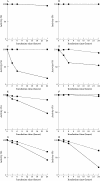
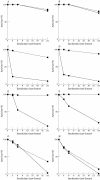
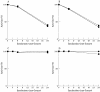
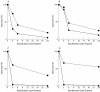
Results: Type MM PGM was inactivated by plasma, whole serum, heated serum, dialysed serum, and serum ultrafiltrate. Inactivation in dialysed serum was reduced by EDTA and largely reversed by thiol agents. Inactivation in serum ultrafiltrate was not prevented by EDTA and only partially reversed by dithiothreitol. The muscle and type BB CK isoenzymes were inactivated in all the tested media. The incubation of human and rabbit skeletal muscle PGM and CK in urate solution showed that urate does not affect mutase activity under conditions that inactivate CK.
Conclusions: These results confirm the mechanisms of CK inactivation proposed by others and show that the type M PGM subunit is inactivated by two different mechanisms, which appear to involve the thiol groups of the enzyme. One mechanism is caused by either a protein component or a protein bound serum component and involves calcium ions and/or another chelatable metal ion. The other mechanism is caused by a lower molecular weight serum component and is metal ion independent.
Figures

Similar articles
-
Phosphoglycerate mutase, 2,3-bisphosphoglycerate phosphatase and creatine kinase activity and isoenzymes in human brain tumours.Br J Cancer. 1997;76(9):1139-49. doi: 10.1038/bjc.1997.525. Br J Cancer. 1997. PMID: 9365161 Free PMC article.
-
Activity of phosphoglycerate mutase and its isoenzymes in serum after acute myocardial infarction.Clin Mol Pathol. 1996 Oct;49(5):M298-300. doi: 10.1136/mp.49.5.m298. Clin Mol Pathol. 1996. PMID: 16696092 Free PMC article.
-
Effects of acrylamide on creatine kinase from rabbit muscle.Int J Biochem Cell Biol. 2001 Nov;33(11):1064-70. doi: 10.1016/s1357-2725(01)00079-6. Int J Biochem Cell Biol. 2001. PMID: 11551822
-
D-2-hydroxyglutaric acid inhibits creatine kinase activity from cardiac and skeletal muscle of young rats.Eur J Clin Invest. 2003 Oct;33(10):840-7. doi: 10.1046/j.1365-2362.2003.01237.x. Eur J Clin Invest. 2003. PMID: 14511354
-
[Creatine kinase and its isozymes].Rinsho Byori. 2001 Nov;Suppl 116:52-61. Rinsho Byori. 2001. PMID: 11797380 Review. Japanese.
Cited by
-
Role of Uric Acid in Semen.Biomolecules. 2018 Jul 31;8(3):65. doi: 10.3390/biom8030065. Biomolecules. 2018. PMID: 30065172 Free PMC article. Review.
-
A Comparative Analysis of the Altered Levels of Human Seminal Plasma Constituents as Contributing Factors in Different Types of Male Infertility.Curr Issues Mol Biol. 2021 Sep 26;43(3):1307-1324. doi: 10.3390/cimb43030093. Curr Issues Mol Biol. 2021. PMID: 34698062 Free PMC article.
-
Metabolomic characterization of semen from asthenozoospermic patients using ultra-high-performance liquid chromatography-tandem quadrupole time-of-flight mass spectrometry.Biomed Chromatogr. 2020 Sep;34(9):e4897. doi: 10.1002/bmc.4897. Epub 2020 Jun 27. Biomed Chromatogr. 2020. PMID: 32428267 Free PMC article.
-
Blood purification with a cytokine adsorber for the elimination of myoglobin in critically ill patients with severe rhabdomyolysis.Crit Care. 2021 Jan 28;25(1):41. doi: 10.1186/s13054-021-03468-x. Crit Care. 2021. PMID: 33509234 Free PMC article.
-
Loss of full-length dystrophin expression results in major cell-autonomous abnormalities in proliferating myoblasts.Elife. 2022 Sep 27;11:e75521. doi: 10.7554/eLife.75521. Elife. 2022. PMID: 36164827 Free PMC article.
References
-
- Fothergill-Gilmore LA, Watson HC. The phosphoglycerate mutases. Adv Enzymol 1989;62:227–313. - PubMed
-
- Mezquita J, Carreras J. Phylogeny and ontogeny of the phosphoglycerate mutases-I. Electrophoretic phenotypes of the glycerate-2,3-P2 dependent phosphoglycerate mutase in vertebrates. Comp Biochem Physiol B Biochem Mol Biol 1981;70B:237–45.
-
- Bartrons R, Carreras J. Purification and characterization of phosphoglycerate mutase isozymes from pig heart. Biochim Biophys Acta 1982;708:167–77. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
