Distinct properties of CRAC and MIC channels in RBL cells
- PMID: 12149283
- PMCID: PMC2234455
- DOI: 10.1085/jgp.20028601
Distinct properties of CRAC and MIC channels in RBL cells
Abstract
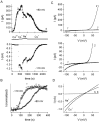
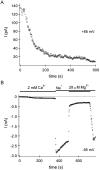
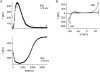
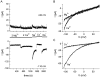
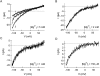
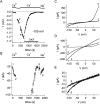
In rat basophilic leukemia (RBL) cells and Jurkat T cells, Ca(2+) release-activated Ca(2+) (CRAC) channels open in response to passive Ca(2+) store depletion. Inwardly rectifying CRAC channels admit monovalent cations when external divalent ions are removed. Removal of internal Mg(2+) exposes an outwardly rectifying current (Mg(2+)-inhibited cation [MIC]) that also admits monovalent cations when external divalent ions are removed. Here we demonstrate that CRAC and MIC currents are separable by ion selectivity and rectification properties: by kinetics of activation and susceptibility to run-down and by pharmacological sensitivity to external Mg(2+), spermine, and SKF-96365. Importantly, selective run-down of MIC current allowed CRAC and MIC current to be characterized under identical ionic conditions with low internal Mg(2+). Removal of internal Mg(2+) induced MIC current despite widely varying Ca(2+) and EGTA levels, suggesting that Ca(2+)-store depletion is not involved in activation of MIC channels. Increasing internal Mg(2+) from submicromolar to millimolar levels decreased MIC currents without affecting rectification but did not alter CRAC current rectification or amplitudes. External Mg(2+) and Cs(+) carried current through MIC but not CRAC channels. SKF-96365 blocked CRAC current reversibly but inhibited MIC current irreversibly. At micromolar concentrations, both spermine and extracellular Mg(2+) blocked monovalent MIC current reversibly but not monovalent CRAC current. The biophysical characteristics of MIC current match well with cloned and expressed TRPM7 channels. Previous results are reevaluated in terms of separate CRAC and MIC channels.
Figures
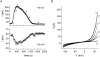



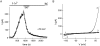


References
-
- Bosteels, S., P. Matejovic, W. Flameng, and K. Mubagwa. 1999. Sodium influx via a non-selective pathway activated by the removal of extracellular divalent cations: possible role in the calcium paradox. Cardiovasc. Res. 43:417–425. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

