Modeling tight junction dynamics and oscillations
- PMID: 12149284
- PMCID: PMC2234459
- DOI: 10.1085/jgp.20028604
Modeling tight junction dynamics and oscillations
Abstract
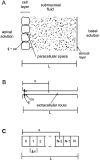
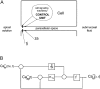
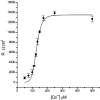
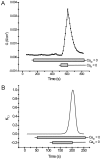
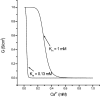
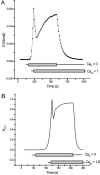
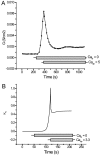
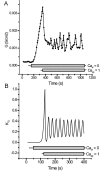
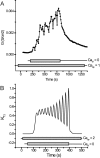
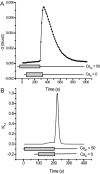
Tight junction (TJ) permeability responds to changes of extracellular Ca(2+) concentration. This can be gauged through changes of the transepithelial electrical conductance (G) determined in the absence of apical Na(+). The early events of TJ dynamics were evaluated by the fast Ca(2+) switch assay (FCSA) (Lacaz-Vieira, 2000), which consists of opening the TJs by removing basal calcium (Ca(2+)(bl)) and closing by returning Ca(2+)(bl) to normal values. Oscillations of TJ permeability were observed when Ca(2+)(bl) is removed in the presence of apical calcium (Ca(2+)(ap)) and were interpreted as resulting from oscillations of a feedback control loop which involves: (a) a sensor (the Ca(2+) binding sites of zonula adhaerens), (b) a control unit (the cell signaling machinery), and (c) an effector (the TJs). A mathematical model to explain the dynamical behavior of the TJs and oscillations was developed. The extracellular route (ER), which comprises the paracellular space in series with the submucosal interstitial fluid, was modeled as a continuous aqueous medium having the TJ as a controlled barrier located at its apical end. The ER was approximated as a linear array of cells. The most apical cell is separated from the apical solution by the TJ and this cell bears the Ca(2+) binding sites of zonula adhaerens that control the TJs. According to the model, the control unit receives information from the Ca(2+) binding sites and delivers a signal that regulates the TJ barrier. Ca(2+) moves along the ER according to one-dimensional diffusion following Fick's second law. Across the TJ, Ca(2+) diffusion follows Fick's first law. Our first approach was to simulate the experimental results in a semiquantitative way. The model tested against experiment results performed in the frog urinary bladder adequately predicts the responses obtained in different experimental conditions, such as: (a) TJ opening and closing in a FCSA, (b) opening by the presence of apical Ca(2+) and attainment of a new steady-state, (c) the escape phase which follows the halt of TJ opening induced by apical Ca(2+), (d) the oscillations of TJ permeability, and (e) the effect of Ca(2+)(ap) concentration on the frequency of oscillations.
Figures
Similar articles
-
Pulses of cell Ca(2+) and the dynamics of tight junction opening and closing.J Membr Biol. 2003 Nov 15;196(2):117-27. doi: 10.1007/s00232-003-0630-2. J Membr Biol. 2003. PMID: 14724748
-
Tight junction dynamics: oscillations and the role of protein kinase C.J Membr Biol. 2000 Nov 15;178(2):151-61. doi: 10.1007/s002320010022. J Membr Biol. 2000. PMID: 11083903
-
Protein kinase inhibitors and the dynamics of tight junction opening and closing in A6 cell monolayers.J Membr Biol. 2001 Nov 15;184(2):185-96. doi: 10.1007/s00232-001-0082-5. J Membr Biol. 2001. PMID: 11719854
-
Implications of AMPK in the Formation of Epithelial Tight Junctions.Int J Mol Sci. 2018 Jul 13;19(7):2040. doi: 10.3390/ijms19072040. Int J Mol Sci. 2018. PMID: 30011834 Free PMC article. Review.
-
Applications of imaging techniques to studies of epithelial tight junctions.Adv Drug Deliv Rev. 2005 Jan 2;57(1):111-21. doi: 10.1016/j.addr.2004.08.004. Adv Drug Deliv Rev. 2005. PMID: 15518924 Review.
Cited by
-
The bacterial virulence factor lymphostatin compromises intestinal epithelial barrier function by modulating rho GTPases.Am J Pathol. 2009 Apr;174(4):1347-57. doi: 10.2353/ajpath.2009.080640. Epub 2009 Mar 12. Am J Pathol. 2009. PMID: 19286565 Free PMC article.
-
Lanthanum effect on the dynamics of tight junction opening and closing.J Membr Biol. 2004 Nov;202(1):39-49. doi: 10.1007/s00232-004-0718-3. J Membr Biol. 2004. PMID: 15702378
-
Colonic epithelial ion transport is not affected in patients with diverticulosis.BMC Gastroenterol. 2007 Sep 23;7:37. doi: 10.1186/1471-230X-7-37. BMC Gastroenterol. 2007. PMID: 17888183 Free PMC article.
-
Pulses of cell Ca(2+) and the dynamics of tight junction opening and closing.J Membr Biol. 2003 Nov 15;196(2):117-27. doi: 10.1007/s00232-003-0630-2. J Membr Biol. 2003. PMID: 14724748
References
-
- Byrne, J.H., and S.G. Schultz. 1988. An Introduction to Membrane Transport and Bioelectricity. Raven Press, New York. 1–232.
-
- Castro, J.A., A. Sesso, and F. Lacaz-Vieira. 1993. Deposition of BaSO4 in the tight junctions of amphibian epithelia causes their opening; apical Ca2+ reverses this effect. J. Membr. Biol. 134:15–29. - PubMed
-
- Cereijido, M., L. González-Mariscal, G. Avila, and R.G. Contreras. 1988. Tight junctions. CRC Crit. Rev. Anat. Sci. 1:171–192. - PubMed
-
- Cereijido, M., L. Shoshani, and R.G. Contreras. 2000. Molecular physiology and pathophysiology of tight junctions. I. Biogenesis of tight junctions and epithelial polarity. Am. J. Physiol. Gastrointest. Liver Physiol. 279:G477–G482. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

