An anti-transferrin receptor-avidin fusion protein exhibits both strong proapoptotic activity and the ability to deliver various molecules into cancer cells
- PMID: 12149472
- PMCID: PMC125019
- DOI: 10.1073/pnas.162362999
An anti-transferrin receptor-avidin fusion protein exhibits both strong proapoptotic activity and the ability to deliver various molecules into cancer cells
Abstract
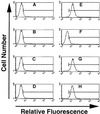
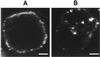
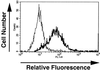
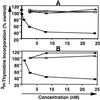
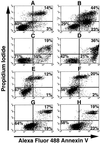
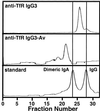
We have developed an antibody fusion protein (anti-rat TfR IgG3-Av) with the ability to deliver different molecules into cancer cells. It consists of avidin genetically fused to the C(H)3 region of a human IgG3 specific for the rat transferrin receptor. It forms strong, noncovalent interactions with biotinylated molecules such as glucose oxidase and beta-galactosidase, and delivers them into the rat myeloma cell line Y3-Ag1.2.3 through receptor-mediated endocytosis. Importantly, the beta-galactosidase retains activity after internalization. Furthermore, we have unexpectedly discovered that anti-rat TfR IgG3-Av, but not a recombinant anti-rat TfR IgG3 or a nonspecific IgG3-Av, possesses proapoptotic activities against Y3-Ag1.2.3 and the rat T cell lymphoma cell line C58 (NT) D.1.G.OVAR.1. These activities were not observed in two rat cell lines of nonhematopoietic lineage (bladder carcinoma BC47 and gliosarcoma 9L). Anti-human TfR IgG3-Av also demonstrated proapoptotic activity against the human erythroleukemia cell line K562. Studies showed that anti-rat TfR IgG3-Av exists as a dimer, suggesting that cross-linking of the surface transferrin receptor may be responsible for the cytotoxic activity. These findings demonstrate that it is possible to transform an antibody specific for a growth factor receptor that does not exhibit inhibitory activity into a drug with significant intrinsic cytotoxic activity against selected cells by fusing it with avidin. The antitumor activity may be enhanced by delivering biotinylated therapeutics into cancer cells. Further development of this technology may lead to effective therapeutics for in vivo eradication of hematological malignancies, and ex vivo purging of cancer cells in autologous transplantation.
Figures

Similar articles
-
Binding specificity and internalization properties of an antibody-avidin fusion protein targeting the human transferrin receptor.J Control Release. 2007 Dec 4;124(1-2):35-42. doi: 10.1016/j.jconrel.2007.08.020. Epub 2007 Aug 25. J Control Release. 2007. PMID: 17884229
-
A human biotin acceptor domain allows site-specific conjugation of an enzyme to an antibody-avidin fusion protein for targeted drug delivery.Biomol Eng. 2005 Feb;21(6):145-55. doi: 10.1016/j.bioeng.2004.10.001. Epub 2004 Dec 9. Biomol Eng. 2005. PMID: 15748688
-
An antibody-avidin fusion protein specific for the transferrin receptor serves as a delivery vehicle for effective brain targeting: initial applications in anti-HIV antisense drug delivery to the brain.J Immunol. 1999 Oct 15;163(8):4421-6. J Immunol. 1999. PMID: 10510383
-
Blood-brain barrier drug delivery of IgG fusion proteins with a transferrin receptor monoclonal antibody.Expert Opin Drug Deliv. 2015 Feb;12(2):207-22. doi: 10.1517/17425247.2014.952627. Epub 2014 Aug 20. Expert Opin Drug Deliv. 2015. PMID: 25138991 Review.
-
Reengineering biopharmaceuticals for targeted delivery across the blood-brain barrier.Methods Enzymol. 2012;503:269-92. doi: 10.1016/B978-0-12-396962-0.00011-2. Methods Enzymol. 2012. PMID: 22230573 Review.
Cited by
-
Two interconvertible folds modulate the activity of a DNA aptamer against transferrin receptor.Mol Ther Nucleic Acids. 2014 Jan 28;3(1):e144. doi: 10.1038/mtna.2013.71. Mol Ther Nucleic Acids. 2014. PMID: 24472870 Free PMC article.
-
A novel transferrin receptor-targeted hybrid peptide disintegrates cancer cell membrane to induce rapid killing of cancer cells.BMC Cancer. 2011 Aug 18;11:359. doi: 10.1186/1471-2407-11-359. BMC Cancer. 2011. PMID: 21849092 Free PMC article.
-
A role for transferrin receptor in triggering apoptosis when targeted with gambogic acid.Proc Natl Acad Sci U S A. 2005 Aug 23;102(34):12095-100. doi: 10.1073/pnas.0406731102. Epub 2005 Aug 15. Proc Natl Acad Sci U S A. 2005. PMID: 16103367 Free PMC article.
-
A Historical Review of Brain Drug Delivery.Pharmaceutics. 2022 Jun 16;14(6):1283. doi: 10.3390/pharmaceutics14061283. Pharmaceutics. 2022. PMID: 35745855 Free PMC article. Review.
-
Insights into the effector functions of human IgG3 in the context of an antibody targeting transferrin receptor 1.Mol Immunol. 2015 Oct;67(2 Pt B):407-15. doi: 10.1016/j.molimm.2015.07.001. Epub 2015 Jul 29. Mol Immunol. 2015. PMID: 26232328 Free PMC article.
References
-
- Larrick J. W. & Cresswell, P. (1979) J. Supramol. Struct. 11, 579-586. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

