Receptor for activated C kinase-1 facilitates protein kinase C-dependent phosphorylation and functional modulation of GABA(A) receptors with the activation of G-protein-coupled receptors
- PMID: 12151513
- PMCID: PMC6758144
- DOI: 10.1523/JNEUROSCI.22-15-06353.2002
Receptor for activated C kinase-1 facilitates protein kinase C-dependent phosphorylation and functional modulation of GABA(A) receptors with the activation of G-protein-coupled receptors
Abstract
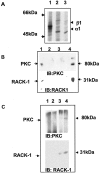
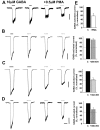
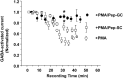
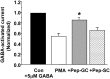
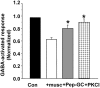
GABA(A) receptors are the principal sites of fast synaptic inhibition in the brain. These receptors are hetero-pentamers that can be assembled from a number of subunit classes: alpha(1-6), beta(1-3), gamma(1-3), delta(1), epsilon, theta;, and pi, but the majority of receptor subtypes is believed, however, to be composed of alpha, beta, and gamma2 subunits. A major mechanism for modulating GABA(A) receptor function occurs via the phosphorylation of residues within the intracellular domains of receptor subunits by a range of serine/threonine and tyrosine kinases. However, how protein kinases are targeted to these receptors to facilitate functional modulation remains unknown. Here we demonstrate that the receptor for activated C kinase (RACK-1) and protein kinase C (PKC) bind to distinct sites on GABA(A) receptor beta subunits. Although RACK-1 is not essential for PKC binding to GABA(A) receptor beta subunits, it enhances the phosphorylation of serine 409, a residue critical for the phospho-dependent modulation of GABA(A) receptor function in the beta1 subunit by anchored PKC. Furthermore, RACK-1 also enhances GABA(A) receptor functional modulation in neurons by a PKC-dependent signaling pathway with the activation of muscarinic acetylcholine receptors (mAChRs). This PKC-dependent modulation of neuronal GABA(A) receptors was mirrored by an increase in the phosphorylation of GABA(A) receptor beta subunits with the activation of mAChRs. Our results suggest a central role for RACK-1 in potentiating PKC-dependent phosphorylation and functional modulation of GABA(A) receptors. Therefore, RACK-1 will enhance functional cross talk between GABA(A) receptors and G-protein-coupled receptors and therefore may have profound effects on neuronal excitability.
Figures



References
-
- Benke D, Fritschy JM, Trzeciak A, Bannwarth W, Mohler H. Distribution, prevalence, and drug binding profile of γ-aminobutyric acid type A receptor subtypes differing in the β-subunit variant. J Biol Chem. 1994;269:27100–27107. - PubMed
-
- Brandon NJ, Smart TG, Moss SJ. Regulation of GABAA receptors by protein phosphorylation. In: Martin DL, Olsen RL, editors. GABA in the nervous system, the view at 50 years. Williams & Wilkins; Baltimore: 2000a. pp. 191–206.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
